✓ 연중무휴 하루 24시간 자동 온라인 주문 처리
✓ 풍부한 지식과 전문성을 갖춘 제품 및 기술 지원
✓ 신속하고 안정적인 (재)주문
QIAcuity EG PCR Kit (1 ml) icon_0368_ls_gen_eco_friendly-s
카탈로그 번호 / ID. 250111
✓ 연중무휴 하루 24시간 자동 온라인 주문 처리
✓ 풍부한 지식과 전문성을 갖춘 제품 및 기술 지원
✓ 신속하고 안정적인 (재)주문
특징
- EvaGreen을 사용한 염료 기반 디지털 PCR 반응용
- 더 많은 샘플을 로딩하기 위한 3배 농축 마스터 믹스
- QIAcuity Nanoplates에서의 미세 유체 사용에 대해 최적화
- REACH 준수
제품 세부 정보
QIAcuity EG PCR Kit에는 QIAcuity Nanoplates에서의 미세 유체 사용에 최적화된 3배 농축, 바로 사용 가능한 마스터 믹스가 포함되어 있습니다. 이 키트는 염료 기반 디지털 PCR의 특이성과 효율성을 개선하여 정확한 정량 분석을 제공합니다. 삽입 염료 EvaGreen은 이중 가닥 DNA에 결합하여 QIAcuity dPCR 기기에서 gDNA 또는 cDNA 측정의 정량적 정확도를 향상합니다.
이 키트는 QIAcuity Digital PCR System 및 QIAcuity Nanoplates와 함께 기능합니다.
제품에 대해 자세히 알아보고 당사의 dPCR 전문가에게 문의하시겠습니까 ? 여기에 정보를 기록하시면 곧 연락을 드리겠습니다.
성능
우월한 성능
EvaGreen 기반 검출을 위한 QIAcuity Master Mixes는 최신 버전의 QIAGEN 고품질 DNA 중합효소를 사용합니다. 새로운 QuantiNova DNA 중합효소와 Nanoplate 미세 유체에 최적화된 QIAGEN의 독점적이고 입증된 완충액 기술의 고유한 조합으로서, 민감도, 재현성 및 효율성 측면에서 매우 일관된 결과가 제공됩니다.
EvaGreen을 사용한 염료 기반 검출
QIAcuity EG PCR Kit의 특별한 마스터 믹스를 사용하여 정확한 이중 가닥 DNA 표적 증폭 및 정량화를 할 수 있습니다. 여기에는 dPCR 분석 및 Nanoplates의 분석 가능한 파티션 계산에 필요한 최적화된 참조 염료가 포함되어 있습니다. 또한 EvaGreen은 동일한 농도에서 SYBR Green보다 더 높은 형광 신호를 제공하고 dPCR에서 최대 증폭 효율, 특이성 및 민감도를 제공합니다.
최대 100시간의 반응 안정성
QIAcuity PCR 혼합물은 후속 반응의 성능을 손상시키지 않고 최대 100시간 동안 30°C에서 보관할 수 있습니다. 냉각제를 사용하지 않고 실온에서 장기간 보관한 후에도 안정성이 뛰어나므로 QIAcuity EG PCR Kit는 처리량이 많은 반응 설정 및 댜량의 플레이트 실험에 이상적입니다.
원리
QIAcuity EG PCR Kit는 새로운 항체 매개 hot-start 작용기전으로 인해 최고 특이성을 가진 cDNA 또는 gDNA 분석을 제공합니다. 저온에서 QuantiNova DNA 중합효소는 QuantiNova Antibody 그리고 복합체를 안정화하는 새로운 첨가제인 QuantiNova Guard에 의해 비활성 상태로 유지됩니다. 이는 hot-start 의 견고성이 향상되고 비특이적으로 어닐링된 프라이머 및 프라이머–이합체(dimer)의 확장을 방지합니다. 95°C까지 온도를 올린 후 2분 이내에 QuantiNova Antibody와 QuantiNova Guard가 변성되고 QuantiNova DNA Polymerase가 활성화되어 PCR 증폭이 가능합니다.
나노플레이트에서의 dPCR 반응 원리는 여기에 설명되어 있습니다.
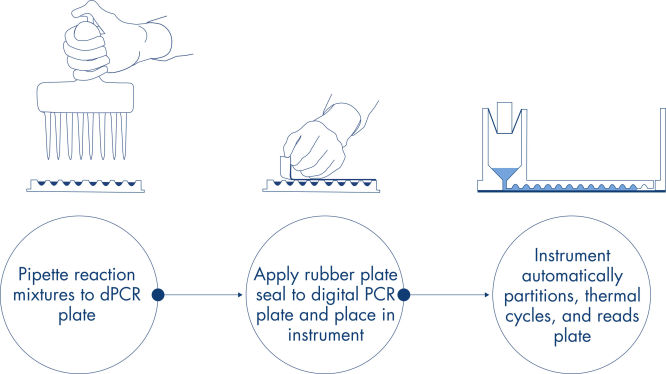
절차
qPCR 실험과 마찬가지로 샘플 준비에는 마스터 믹스, 프로브 및 프라이머를 96웰 또는 24웰 나노플레이트로 옮긴 다음 샘플을 추가하는 작업이 포함됩니다. 이 시스템은 분획, 열순환(thermocycling) 및 이미징을 하나의 완전 자동화 기기로 통합하여 사용자가 2시간 이내에 샘플에서부터 결과를 얻을 수 있습니다. Suite 소프트웨어에서 분석을 수행할 수 있으며, 표적 서열의 마이크로리터당 복제 농도뿐만 아니라 양성 검체나 NTC와 같은 품질 관리도 제공됩니다. 이 분석은 또한 동일한 LAN(근거리 통신망) 내의 원격 컴퓨터로 확장될 수 있습니다.
응용 분야
QIAcuity EG PCR Kit는 QIAcuity Digital PCR System 및 QIAcuity Nanoplates와 함께 다음을 포함한 애플리케이션에 사용될 cDNA 표적 및 gDNA의 정량 분석을 가능하게 합니다.
- 희귀 돌연변이 검출
- 복제 수 변이 분석
- 유전자 발현 분석
- 병원체 검출
- 유전형 분석
- miRNA 연구
지원되는 데이터 및 수치
간단하고 신속한 플레이트 기반 워크플로우