✓ Procesamiento automático sin interrupción de pedidos en línea
✓ Servicio técnico y para productos experto y profesional
✓ Realización y repetición de pedidos rápidas y fiables
QIAcuity EG PCR Kit (1 ml) icon_0368_ls_gen_eco_friendly-s
N.º de cat. / ID. 250111
✓ Procesamiento automático sin interrupción de pedidos en línea
✓ Servicio técnico y para productos experto y profesional
✓ Realización y repetición de pedidos rápidas y fiables
Características
- Para reacciones de PCR digital basadas en colorante en las que se utiliza EvaGreen
- Mezcla maestra con concentración de 3× para cargar más muestra
- Optimizado para el uso de microfluidos en QIAcuity Nanoplates
- Conforme a la norma REACH
Detalles del producto
El QIAcuity EG PCR Kit contiene mezcla maestra lista para usar con concentración de 3× optimizada para el uso de microfluidos en las QIAcuity Nanoplates. El kit mejora la especificidad y la eficacia de la PCR digital basada en colorante para ofrecer análisis de cuantificación exactos. Un colorante de intercalado EvaGreen se une al ADN de doble cadena y mejora la exactitud cuantitativa de las mediciones de ADNg o ADNc en los instrumentos de dPCR QIAcuity.
El kit funciona junto con el QIAcuity Digital PCR System y las QIAcuity Nanoplates.
¿Le gustaría obtener más información sobre el producto y que uno de nuestros especialistas en dPCR se ponga en contacto con usted? Regístrese aquí y nos pondremos en contacto con usted en breve.
Rendimiento
Rendimiento superior
Las QIAcuity Master Mixes para la detección basada en EvaGreen utilizan las versiones más recientes de ADN polimerasa de alta calidad de QIAGEN. La combinación única de la patentada tecnología de tampón de eficacia comprobada de QIAGEN optimizada para microfluidos de nanoplacas, junto con el nuevo QuantiNova DNA Polymerase permite obtener resultados uniformes en lo que respecta a sensibilidad, reproducibilidad y eficacia.
Detección basada en colorante con EvaGreen
La mezcla maestra especial del QIAcuity EG PCR Kit permite la amplificación y cuantificación exactas de blanco de ADN de doble cadena. Incluye un colorante de referencia optimizado necesario para el análisis de dPCR y el recuento de divisiones analizables en las Nanoplates. Además, EvaGreen proporciona una señal de fluorescencia más elevada que SYBR Green en las mismas concentraciones y ofrece máxima amplificación, eficiencia, especificidad y sensibilidad en dPCR.
Estabilidad en la reacción de hasta 100 horas
Las mezclas para PCR QIAcuity pueden almacenarse a una temperatura de 30 °C durante un máximo de 100 horas sin que se altere el rendimiento de las reacciones posteriores. La excelente estabilidad, incluso tras un almacenamiento prolongado a temperatura ambiente sin el uso de ningún agente refrigerante hace que el QIAcuity EG PCR Kit sea ideal para la configuración de reacciones de alto rendimiento y la manipulación de pilas de placas.
Principio
El QIAcuity EG PCR Kit proporciona análisis de ADNc o ADNg con máxima especificidad gracias a su novedoso mecanismo de inicio en caliente mediado por anticuerpos. A bajas temperaturas, el QuantiNova DNA Polymerase se mantiene en un estado inactivo por acción del anticuerpo QuantiNova Antibody y un nuevo aditivo, QuantiNova Guard, que estabiliza el complejo. De esta manera, se mejora la astringencia del inicio en caliente y previene la extensión de oligonucleótidos alineados de manera no específica y de dímeros de oligonucleótidos. A los 2 minutos de haber alcanzado una temperatura de 95 °C, el QuantiNova Antibody y el QuantiNova Guard se desnaturalizan y se activa la QuantiNova DNA Polymerase, lo que permite la amplificación por PCR.
El principio de la reacción de dPCR en las nanoplacas se describe aquí.
Procedimiento
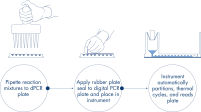
Al igual que en los experimentos de qPCR, la preparación de las muestras incluye la transferencia de mezcla maestra, sondas y cebadores a una nanoplaca de 96 o 24 pocillos, seguida de la adición de las muestras. El sistema integra funciones de división en partes, termociclado y obtención de imágenes en un solo instrumento completamente automatizado que permite a los usuarios obtener los resultados de las muestras en menos de 2 horas. Es posible realizar un análisis en el paquete de software, que proporciona la concentración en copias por microlitro de la secuencia de blancos y también para el control de calidad como muestras positivas o NTC. Este análisis también se puede extender a ordenadores remotos dentro de la misma red de área local (LAN).
Aplicaciones
El QIAcuity EG PCR Kit, junto con el QIAcuity Digital PCR System y las QIAcuity Nanoplates, permiten el análisis cuantitativo de blancos de ADNc y ADNg para utilizarlas en aplicaciones, entre las que se incluyen:
- Detección de mutaciones poco frecuentes
- Análisis de variación en el número de copias
- Análisis de expresión génica
- Detección de microrganismos patógenos
- Genotipado
- Investigación sobre miARN
Datos y cifras de respaldo
Flujo de trabajo simple y rápido basado en la placa