✓ オンライン注文による24時間年中無休の自動処理システム
✓ 知識豊富で専門的な製品&テクニカルサポート
✓ 迅速で信頼性の高い(再)注文
QuantiFast Pathogen RT-PCR +IC Kit (400)
Cat. No. / ID: 211454
✓ オンライン注文による24時間年中無休の自動処理システム
✓ 知識豊富で専門的な製品&テクニカルサポート
✓ 迅速で信頼性の高い(再)注文
特徴
- 病原体ターゲットとインターナルコントロールを同時に検出
- 5x マスターミックスでサンプル添加量を増加でき感度を増大
- 陰性結果の明確化により、病原体の新規アッセイ系を簡便に構築
- 低い陽性シグナルも明確に検出
- スタンダードおよび高速サイクラーに対応する迅速な共通プロトコール
製品詳細
QuantiFast Pathogen +IC Kits は、配列特異的なプローブを用いたリアルタイムPCR あるいは1 ステップRT-PCR により、病原体核酸を高感度かつ高速に検出するために特化されています。偽陰性を排除した確実性の高い系を実現するために、最高4種類の研究対象の病原体ターゲット(ウイルス、バクテリア、真菌など)とインターナルコントロール(IC)をリアルタイムに検出するための試薬が各キットに入っています。キットフォーマットは2種類あります:Internal RNA Controlテンプレートとこれを検出するInternal Controlプライマー/プローブセットが入ったウイルスRNA 検出用QuantiFast Pathogen RT-PCR +IC Kit、あるいはInternal DNA Controlテンプレートとこれを検出するInternal Control プライマー/プローブセットが入ったウイルス/バクテリア/真菌DNA 検出用 QuantiFast Pathogen PCR +IC Kit。両キットともに2種類の濃度のROX(チューブ)がキットに入っているので、どのようなリアルタイムサイクラーにも使用可能です。マスターミックスは2~8 ℃で保存でき、簡便に取り扱えます。
パフォーマンス
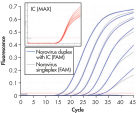
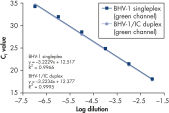
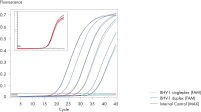
QuantiFast Pathogen +IC Kitsを用いたマルチプレックスアッセイは、感度を損なうことなく複数のウイルスRNAやDNA ターゲットと添付のインターナルコントロールを同時に幅広い範囲で正確に検出できます(図” Rotor-Gene Qでノロウイルスの高感度検出”および“ Singleplexと Duplex検出で高い直線性と精度を実現”)。本プロトコールはほとんどのサイクラーで迅速なサイクリング実験を高い信頼性で行なうために開発されました(図“ Rotor-Gene QでBHV-1の高感度検出”および“ ABI 7500でBHV-1の高感度検出”)。QuantiFast Pathogen +IC Kitsを用いて病原体ターゲットとインターナルコントロールの増幅を組み合わせることにより、陰性結果を明確に判定できるので、確実な病原体検出用のワークフローを実現します(図“ 陰性結果の正確な判定”)。
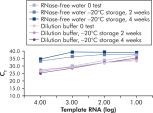
キットに同梱のQuantiTect Nucleic Acid Dilution Bufferは、希釈および反応セットアップ中にRNAやDNAスタンダードを安定化し、チューブやピペットチップの様なプラスチック表面への核酸の吸着を防ぎます。このバッファーによりウイルス核酸の定量に用いるスタンダードを正確に希釈でき、広範囲のCT 値において高い直線性が得られます。本バッファーはスタンダードの分解を防ぎ、長期保存を可能にします(図 “ RNA スタンダードの正確な希釈および保存”)。
図参照
原理
陰性結果を明確に判定でき確実性の高い系を実現するため、インターナルコントロールと対象とする病原体の標的遺伝子を同時に検出するマルチプレックスリアルタイム検出用試薬が各QuantiFast Pathogen +IC Kitに入っています。別々の反応ではなく同一反応内でコントロール遺伝子と標的遺伝子を増幅することで、マニュアルでの作業によるエラーが最小限に抑えられ、遺伝子定量の信頼性が高くなります。
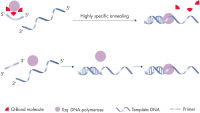
QuantiFast Pathogen +IC Kitsは、最初の実験でマルチプレックスリアルタイムPCRや1ステップ RT-PCRによる高感度で迅速な病原体核酸検出を実現します(フローチャート“ QIAGEN Multiplex Kit”)。至適化されたマスターミックスにより、マルチプレックス反応でのPCR産物をシングル増幅反応に相当するPCR産物と同様の効率および感度で確実に増幅します。特別に開発された高速PCR用バッファーは、変性、アニーリングおよびエクステンション時間も顕著に短縮する斬新なQ-Bondを含有しています(図“ プライマーの高速なアニーリング”)。画期的な合成添加剤Factor MPとK+およびNH4+のイオン配合比により、プライマーとプローブが核酸テンプレートに効率的かつ安定してアニーリングし、高いPCR効率と感度を実現します(図“ ユニークなPCRバッファー”)。さらにSensiscript Reverse Transcriptaseのユニークな組成はウイルスRNAの高感度な逆転写を実現し、HotStarTaq Plus DNA Polymeraseは厳密なホットスタートにより非特異的な産物の生成を回避します。
| 成分 | 特長 | 利点 | |
|---|---|---|---|
| 5x QuantiFast Pathogen PCR Master Mix | 高濃度マスターミックス | 高感度な病原体検出用に高濃度で至適化済み | テンプレート量を増やしアッセイ感度の増大を実現 |
| HotStarTaq Plus DNA Polymerase | 95℃、5分の活性化 | 室温で定量PCRのセットアップ | |
| QuantiFast Pathogen Buffer | NH4+とK+イオンの配合バランス | 特異性の高いプライマーのアニーリングで信頼性の高いPCR結果 | |
| 合成添加剤Factor MP | 同一チューブ中の最高4遺伝子を高い信頼性でマルチプレックス解析 | ||
| ユニークな添加剤Q-Bond | PCR反応時間が短縮されるため迅速に結果が得られ、1日あたりの反応数を増やせる | ||
| Internal Control Assay | インターナルコントロールテンプレート | QuantiFast Pathogen PCR +IC KitのInternal Control DNAテンプレート | 異なる病原体アッセイとともに使用できるユニバーサルなDNA増幅コントロール |
| QuantiFast Pathogen RT-PCR +IC KitのInternal Control RNA | 異なる病原体アッセイとともに使用できるユニバーサルなRNA増幅コントロール | ||
| Internal Control Assay | MAX(HEX、VICなどに相当)で標識したプレミックスのプライマー/プローブセット(TaqManプローブ) | 標的病原体に対するプライマーを阻害しない | |
| 追加のキット成分 | QuantiFast Pathogen RT Mix* | ユニークな組成のSensiscript Reverse Transcriptase | 病原体RNAの高感度検出用に至適化済み |
| ROX Dye Solution | Applied Biosystems 7500リアルタイムPCRシステムでの蛍光補正用にpassive reference dye が別途チューブ包装。オプション:Agilent、Stratagene 装置で使用可能 | ROX色素が必要なサイクラーで正確な定量。どのリアルタイムサイクラーでもPCRを妨害しない | |
| High-ROX Dye Solution | Applied Biosystems 7900およびStepOneリアルタイムPCR装置での蛍光補正用にpassive reference dyeが別途チューブ包装 | ||
| QuantiTect Nucleic Acid Dilution Buffer | 核酸スタンダードの希釈および保存に最適な独自のバッファー組成 | 希釈や反応セットアップ中のRNAやDNAスタンダードを安定化し、チューブやピペットチップのようなプラスチック表面への核酸の吸着を防止 |
図参照
操作手順
QuantiFast Pathogen +IC Kitsは研究対象の病原体とインターナルコントロールの検出を簡単な操作で実現します。本キットは即使用可能なマスターミックスを含み、ウイルスRNA(1ステップRT-PCR)あるいはウイルス/バクテリア/真菌DNA(PCR)のリアルタイム検出を実現します。反応条件やサイクリング条件の至適化は不要です。マスターミックスに病原体アッセイ(プライマーとプローブ)とInternal Control Assay およびInternal Control DNA/RNAをミックスするだけです。あるいは、Internal Control DNA/RNAを核酸精製プロセスで添加した場合は、Internal Control DNA/RNAの代わりにRNase フリー水を反応ミックスに添加します。その後、DNA あるいはRNA テンプレートを添加し、サイクラーで反応を開始します。ハンドブックにはTaqMan プローブ用に至適化されたプロトコールが入っており、様々なサイクラーで使用可能です。また推奨する色素の組み合わせも記載されています。
各QuantiFast Pathogen +IC Kits に入っている Internal Control Assay およびInternal Control DNA/RNA は、反応ミックスに直接添加して増幅コントロールとして使用します。あるいは核酸精製プロセスおよび増幅の両方をコントロールするために、インターナルコントロールを核酸精製プロセスで添加することも可能です。精製プロセスでインターナルコントロールを添加する場合は、高濃度のInternal Control DNA あるいはRNA(High conc.)を別途注文できます(図“ QIAGEN Internal Control”)。
図参照
アプリケーション
裏付けデータと数値
Correct interpretation of negative results.

Specifications
| Features | Specifications |
|---|---|
| ApplicationsJA | Pathogen Detection: Real-time PCR of viral, bacterial or fungal DNA (QuantiFast Pathogen PCR +IC Kit) or one-step RT-PCR for detection of viral RNA (QuantiFast Pathogen RT-PCR +IC Kit) |
| Sample/target type | QuantiFast Pathogen PCR +IC Kit: viral, bacterial or fungal DNA; QuantiFast Pathogen RT-PCR +IC Kit: viral RNA |
| Single or multiplex | Duplex |
| Reaction type | Real-time PCR or one-step RT-PCR including of an internal control (IC) |
| Real-time or endpoint | Real-time |
| SYBR Green I or sequence-specific probes | Sequence-specific probes |
| Thermal cycler | For most standard and fast real-time cylcers compatible with duplex PCR/RT-PCR, e.g. Rotor-Gene Q or cyclers from Agilent, Applied Biosystems, BioRad, Roche |
| With or without ROX | Master Mix is provided without ROX dye, but 2 separate ROX solutions are included: High-ROX Dye Solution for use with ABI cyclers except ABI 7500, ROX Dye Solution (low ROX conc.) for use with ABI 7500 and other suppliers |