QIAquick Gel Extraction Kit – zur DNA-Extraktion aus Gelen
Zur Gelextraktion/Reinigung von bis zu 10 µg DNA (70 bp bis 10 kb) aus Gel oder enzymatischen Reaktionen
Zur Gelextraktion/Reinigung von bis zu 10 µg DNA (70 bp bis 10 kb) aus Gel oder enzymatischen Reaktionen
✓ Automatische Verarbeitung von Online-Bestellungen 24/7
✓ Sachkundiger und professioneller technischer und Produkt-Support
✓ Schnelle und zuverlässige (Nach-)Bestellung
Kat.-Nr. / ID. 28704
✓ Automatische Verarbeitung von Online-Bestellungen 24/7
✓ Sachkundiger und professioneller technischer und Produkt-Support
✓ Schnelle und zuverlässige (Nach-)Bestellung
Das QIAquick Gel Extraction Kit enthält Spin-Säulen, Puffer und Sammelröhrchen zur Silikamembran-basierten Aufreinigung von DNA-Fragmenten aus Gel (Schnitte von bis 400 mg) und enzymatischen Reaktionen. DNA der Größe 70 bp bis 10 kb wird durch einfaches und schnelles Verfahren mit den Schritten „Binden-Waschen-Eluieren“ mit einem Elutionsvolumen von 30–50 µl aufgereinigt. Ein integrierter pH-Indikator erlaubt die einfache Bestimmung des optimalen pH-Werts für die DNA-Bindung an die Spin-Säule. Das QIAquick PCR & Gel Cleanup Kit enthält auch Puffer für die Aufreinigung von PCR-Produkten > 100 bp und DNA von bis zu 10 kb. Das Verfahren kann auf dem QIAcube Connect vollständig automatisiert werden.
Für optimale Ergebnisse wird empfohlen, dieses Produkt zusammen mit QIAvac 24 Plus zu verwenden.
Das QIAquick Gel Extraction Kit ermöglicht die Entfernung von Nukleotiden, Enzymen, Salzen, Agarose, Ethidiumbromid und anderen Verunreinigungen aus Proben und gewährleistet eine DNA-Rückgewinnung von bis zu 80 % (siehe Abbildung „ Hohe Rückgewinnung aus Gelen“). Mithilfe einer Mikrozentrifuge oder eines Vakuumverteilers wird aus 1–24 Proben DNA im Bereich von 70 bp bis 10 kb aufgereinigt. Aufgereinigte DNA kann beispielsweise zur Sequenzierung verwendet werden (siehe Abbildung „ Zuverlässige Sequenzierung nach Gelextraktion“). DNA-Fragmente kleiner als 70 bp oder größer als 10 kb sollten mit dem QIAEX II Gel Extraction System extrahiert werden.
Die QIAquick PCR-Aufreinigung entfernt Primer, Nukleotide, Enzyme, Mineralöl, Salze und andere Verunreinigungen aus DNA-Proben (siehe Abbildung „ Vollständige Primerentfernung nach PCR“). Mithilfe einer Mikrozentrifuge oder eines Vakuumverteilers wird DNA im Bereich von 100 bp bis 10 kb aufgereinigt.
QIAquick Kits enthalten eine Silika-Membraneinheit für die Bindung von DNA in Hochsalzpuffer und die Elution mit Niedrigsalzpuffer oder Wasser. Durch die Aufreinigung werden Primer, Nukleotide, Enzyme, Mineralöl, Salze, Agarose, Ethidiumbromid und andere Verunreinigungen aus DNA-Proben entfernt (siehe Abbildung „ Hohe Rückgewinnung aus Gelen“). Die Silika-Membrantechnologie beseitigt die Probleme und Schwierigkeiten, die mit losen Harzen und Suspensionen verbunden sind. Spezialisierte Bindepuffer sind für spezifische Anwendungen optimiert und fördern die selektive Adsorption von DNA-Molekülen innerhalb bestimmter Größenbereiche.
Der mitgelieferte Gel-Ladefarbstoff ermöglicht eine schnellere und bequemere Verarbeitung und Analyse der Proben. Der GelPilot Loading Dye enthält drei Farbstoffe zur Nachverfolgung (Xylencyanol, Bromphenolblau und Orange G), um die Optimierung der Agarosegel-Laufzeit zu erleichtern und zu verhindern, dass kleinere DNA-Fragmente zu weit migrieren (siehe Abbildung „GelPilot Loading Dye“).
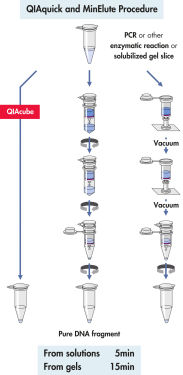
Das QIAquick System nutzt ein einfaches Verfahren mit den Schritten „Binden-Waschen-Eluieren“ (siehe Flussdiagramm „ QIAquick- und MinElute-Verfahren“). Die Gelschnitte werden in einem Puffer aufgelöst, der einen pH-Indikator enthält und die einfache Bestimmung des optimalen pH-Werts für die DNA-Bindung ermöglicht. Die Mischung wird auf die QIAquick Spin Column aufgetragen (siehe Abbildung „ pH-Indikatorfarbstoff“). Die Nukleinsäuren werden unter den Hochsalzbedingungen des Puffers an die Silikamembran adsorbiert. Verunreinigungen werden durch Waschen entfernt, und die reine DNA wird in einem kleinen Volumen des mitgelieferten Niedrigsalzpuffers oder mit Wasser eluiert und kann direkt für alle nachfolgenden Anwendungen genutzt werden.
QIAquick Spin Columns sind so konzipiert, dass sie zwei praktische Optionen für die Handhabung bieten. Die Spin-Säulen passen in eine herkömmliche Tisch-Mikrozentrifuge oder auf jeden Vakuumverteiler mit Luer-Anschlüssen, beispielsweise QIAvac 24 Plus mit QIAvac Luer Adapters. Das QIAquick Gel Extraction Kit sowie andere QIAGEN Kits auf Basis von Spin-Säulen können auf dem QIAcube Connect vollständig automatisiert werden, was eine höhere Produktivität und die Standardisierung der Ergebnisse ermöglicht (siehe Abbildungen „Handhabungsoptionen für Spin-Säulen A, B, C und D“ und „ QIAcube Connect“).
Die mit dem QIAquick System aufgereinigten DNA-Fragmente können direkt für alle Anwendungen verwendet werden, darunter Sequenzierung, Ligation und Transformation, Restriktionsverdau, Markierung, Mikroinjektion, PCR und In-vitro-Transkription.
| Eigenschaften | Spezifikationen |
|---|---|
| Binding capacity | 10 µg |
| Format | Röhrchen |
| Fragment size | 70 bp – 10 kb |
| Recovery: oligonucleotides dsDNA | Rückgewinnung: dsDNA-Fragmente |
| Processing | Manuell |
| Removal <10mers 17–40mers dye terminator proteins | Entfernung von < 10meren |
| Elution volume | 30–50 µl |
| Technology | Silika-Technologie |
| Sample type: applications | DNA: PCR-Reaktionen |