QuantiFast Multiplex RT-PCR Kits
配列特異的なプローブを用いた遺伝子発現解析用高速1ステップマルチプレックスリアルタイム定量RT-PCR
配列特異的なプローブを用いた遺伝子発現解析用高速1ステップマルチプレックスリアルタイム定量RT-PCR

Cat. No. / ID: 204854
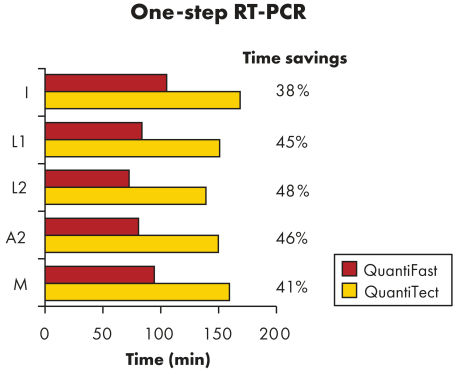
QuantiFast Multiplex RT-PCR KitはRT-PCR時間を最大50%まで短縮できるため、結果がより速く得られます(図 " RT-PCR時間を顕著に短縮")。従って、サンプル数を大幅に増やしたり、他の実験者と1 台のサイクラーを効率的に共有できます。別々の反応ではなく同一反応内でコントロール遺伝子と標的遺伝子を増幅することで、マニュアルでの作業によるエラーが最小限に抑えられ、遺伝子定量の信頼性が増大します(図" 信頼性のある相対定量")。QuantiFast Multiplex RT-PCR Kitに添付の特化されたマスターミックスにより、迅速なマルチプレックス反応のセットアップができ、singleplex RT-PCRデータに匹敵するマルチプレックスRT-PCR データを提供し、初めての実験でも良好な結果が得られます(図 " triplex とsingleplex RT-PCR で同等の結果を実現")。
QuantiFast Multiplex RT-PCR Kit を用いると、ターゲット量のわずかな差異を明確に判別できます。本キットは、2 倍希釈しかしていないテンプレートを用いた場合でも、テンプレート中で量が大きく異なるターゲットを正確に定量できます。最高4 種類のターゲットのマルチプレックスリアルタイムPCR が性能を損なうことなく高速で行なえます(図 " 卓越した感度の4-plex RT-PCR")。
QuantiFast Multiplex RT-PCR Kitsでは最適化実験の必要はなく、標準サイクラーまたは高速サイクラーのどちらでも幅広いダイナミックレンジの高感度かつ迅速な結果が得られます(フローチャート " QIAGEN multiplex kits")。特別に開発された高速PCR 用バッファーは斬新なQ-Bondを含有し、変性、アニーリングおよびエクステンション時間を顕著に短縮します(図" プライマーの高速なアニーリング")。
別々の反応ではなく同一反応内でコントロール遺伝子と標的遺伝子を増幅することで、マニュアルでの作業によるエラーが最小限に抑えられ、遺伝子定量の信頼性が高くなります。QuantiFast Multiplex RT-PCR Buffer のK+ およびNH4+ イオン配合比により、プライマーの特異的なアニーリングを促進され、さらにユニークなFactor MP が特異的に結合したプライマーを安定化します(図 " ユニークなPCRバッファー")。さらに、至適化済み逆転写酵素ミックスにより、わずか20分で効率的なにcDNAを合成できます。また、HotStarTaq Plus DNA Polymeraseは厳密なホットスタートが可能で、非特異的産物の形成を防ぎます。
| 成分 | 特長 | 利点 |
|---|---|---|
| HotStarTaq Plus DNA Polymerase | 95℃、5分の活性化 | 室温での定量PCRのセットアップ |
| QuantiFast Multiplex RT-PCR Buffer | NH4+ イオンおよび K+ イオンの配合バランス | 特異性の高いプライマーのアニーリングで信頼性の高いPCR結果 |
| 合成添加剤Factor MP | 同一チューブで4遺伝子まで信頼できるマルチプレックス解析が可能 | |
| ユニークなQ-Bond を含む | PCR反応時間が短縮されるため迅速に結果が得られ、1日あたりの反応数を増やせる | |
| ROX色素† | Applied Biosystems製装置およびAgilent製装置での蛍光シグナルを補正 | ROXが必要なサイクラーで正確な定量。他のリアルタイムサイクラーでの反応を妨害しない |
| QuantiFast RT Mix | RNAへのアフィニティーが高い逆転写酵素の特殊な配合 | 複雑な二次構造のRNAでも、わずか20分で転写可能 |
QuantiFast Multiplex RT-PCR Kit は反応条件やサイクリング条件の至適化が不要で即使用可能なマスターミックスです。テンプレートRNA、プライマープローブセットをマスターミックスに加えハンドブックのプロトコール通りに操作するだけで、どのリアルタイムサイクラーでも迅速に信頼性の高い結果が得られます。マスターミックス中にROX passive reference dye が入ったキットまたは入っていないキットをお求めいただけますので、実質的にあらゆるリアルタイムサイクラーで使用できます(表を参照)。ROX濃度が至適化されているため、コピー数が少ない場合の検出でも自動データ解析を行なえます。
| ROX 色素 | キット | 対応するサイクラー |
|---|---|---|
| マスターミックスに添加済み | QuantiFast Multiplex RT-PCR Kit | Applied Biosystems 7500以外のApplied Biosystemsの全てのサイクラー |
| 別チューブで添付 | QuantiFast Multiplex RT-PCR +R Kit | Applied Biosystems 7500 および Bio-Rad、Cepheid、Eppendorf、QIAGEN、Roche、Agilent、その他の会社のサイクラー |
| Features | Specifications |
|---|---|
| ApplicationsJA | Real-time quantification of RNA targets in a multiplex format |
| Reaction type | Real-time one-step RT-PCR |
| Single or multiplex | Multiplex |
| SYBR Green I or sequence-specific probes | Sequence-specific probes |
| Real-time or endpoint | Real-time |
| Thermal cycler | Real-time cyclers dedicated for multiplex PCR (e.g., most Applied Biosystems real-time PCR cyclers, Roche LightCycler 480, and Bio-Rad iCycler iQ) |
| Sample/target type | RNA |
| With or without ROX | Available with ROX in master mix or with ROX as separate vial |