- Experiment Configurator
- Discovery & Translational Research
- Diagnostics & Clinical Research
- Human ID & Forensics
- Next-Generation Sequencing
- Instruments & Automation
- Informatics & Data
- Services
- OEM by QIAGEN
- Top Sellers
- New Solutions
Enzymes for molecular biology
Explore high-quality enzymes; now available as individual products.
Products and tools for your targets
Explore targets and pathways in their scientific context, find and customize products to study them, analyze data and plan follow-up studies – all in GeneGlobe.
QuantiFERON-TB Gold Plus
Detect TB infection with confidence.

✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
Cat. No. / ID: 250111
✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
Features
- EvaGreenを使用した蛍光色素ベースのデジタルPCR反応
- より多くのサンプルをロードできる3倍濃縮マスターミックス
- QIAcuity Nanoplatesでのマイクロ流体の使用に最適
- REACH準拠
Product Details
QIAcuity EG PCR Kitには、QIAcuity Nanoplatesでのマイクロ流体の使用に最適なすぐに使える3xマスターミックスが含まれています。このキットは、蛍光色素を使用するデジタルPCRの特異性と効率を向上させ、正確な定量分析を提供します。インターカレート色素EvaGreenは二本鎖DNAに結合し、QIAcuityデジタルPCR装置でのgDNAまたはcDNA測定の定量精度を高めます。
本キットはQIAcuity Digital PCR SystemおよびQIAcuity Nanoplatesと組み合わせて使用します。
製品について詳しくお知りになりたい場合は、弊社のdPCRスペシャリストから連絡を差し上げますので、こちらからサインインしてください。
Performance
卓越した性能
検出色素EvaGreenを使用したQIAcuityマスターミックスは、QIAGENの最新の高品質DNAポリメラーゼを採用しています。新しいQuantiNova DNA Polymeraseとナノプレートマイクロ流体に最適化されたQIAGEN独自の実績のあるバッファーバッファーテクノロジーの組み合わせは、感度、再現性、効率において一貫性の高い結果を提供します。
蛍光色素EvaGreenによる検出
QIAcuity EG PCR Kitの特別なマスターミックスは、ターゲットの二本鎖DNAを正確に増幅および定量できます。キットには、dPCR解析とナノプレート上の解析可能な分画の計数に必要とされる最適なリファレンス色素が含まれます。さらに、EvaGreenはSYBR Greenよりも同一濃度における蛍光強度が高く、dPCRの増幅効率、特異性、および感度を最大限に高めます。
最大100時間までの反応安定性
QIAcutiy PCRミックスは、その後の反応性能を損なうことなく、30°Cで最大100時間保存できます。この優れた安定性は、冷却剤を使用せずに室温で長期間保管できるため、QIAcuity EG PCR Kitはハイスループット反応でのサンプルセットアップやプレートスタッカーでの取り扱いに理想的です。
Principle
QIAcuity EG PCR Kitは、新しい抗体を使ったホットスタート機構により、cDNAまたはgDNA解析を高い特異性をもって実現します。QuantiNova DNA Polymeraseは、QuantiNova抗体および複合体を安定化させる新しい添加剤QuantiNova Guardによって低温で不活化状態を維持します。これにより、ホットスタートの厳密性が改善されて、非特異的にアニーリングしたプライマーおよびプライマーダイマーの伸長を抑制します。温度を95°Cに上げると、2分以内にQuantiNova抗体とQuantiNova Guardは変性し、QuantiNova DNA Polymeraseが活性化されてPCR増幅が可能になります。
ナノプレートでのdPCRの原理はこちらで説明しています。
Procedure
qPCR実験と同様に、サンプル調製では、マスターミックス、プローブ、およびプライマーを96または24ウェルナノプレートへ加え、その後サンプルを加えます。このシステムは、サンプル分配、熱サイクル、イメージングを1つに統合した完全自動化装置です。2時間未満でサンプルの結果が得られます。Software Suiteは、ターゲットシーケンスのマイクロリットルあたりのコピー数で濃度を測定するだけでなく、陽性サンプルやNTCなどの品質コントロールも解析できます。この解析は、同じローカルエリアネットワーク(LAN)内の離れた場所のコンピューターでも行うことができます。
Applications
QIAcuity EG PCR Kitは、QIAcuity Digital PCR SystemおよびQIAcuity Nanoplatesと組み合わせて、次のアプリケーションでのcDNAやgDNAターゲットの定量解析に利用できます。
- 希少突然変異の検出
- コピー数多型解析
- 遺伝子発現解析
- 病原体検出
- 遺伝子型決定
- miRNA研究
Supporting data and figures
プレート方式のシンプルで迅速なワークフロー
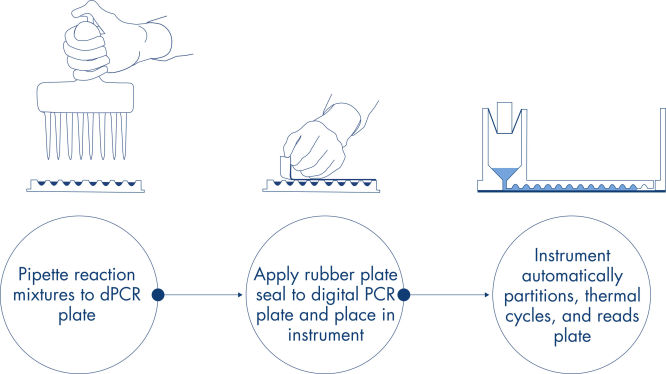
Resources
The Volume Precision Factor (VPF) offers a unique feature to secure precision of concentration results obtained from a QIAcuity dPCR run.
In general, Nanoplates provide partitions of fixed sizes that enable a very precise way of sample concentration calculation. Potential variation of partition sizes in Nanoplate batches, caused by different microstructure molding forms, can be addressed by applying the batch specific VPF. Furthermore, the VPF includes well-specific volume information and therefore further increases precision of concentration calculation in each well of the Nanoplates.
After downloading and updating the VPF file within the QIAcuity Software Suite, the VPF is applied automatically to the analysis of a corresponding Nanoplate batch. The VPF file includes information from all available microstructure molding forms and connected Nanoplate batches. It will be stored on the PC where the QIAcuity Software Suite is installed.
Required QIAcuity Software Suite version: Version 1.2 or higher.
Version 2.1
Version 2.1
Beside of usability improvements and bug fixes, the new features address GMP requirements by offering additional support for 21 CFR part 11 compliance, copy number variation (CNV) analysis, support of future Nanoplates, and improved 1D scatterplot views.
For more information, please refer to the Release Note: QIAcuity Software Suite, Version 2.1
SHA1 checksum: 6D51E273B26FA7D7E0EDA843CB3DCD74B38FA5B1
The QIAcuity Control Software is an integral part of the QIAcuity instrument. It offers a GUI (graphical user interface) for basic functionalities such as plate setup, changing the order of plates to be processed, and monitoring the status of runs in real time. After a run is completed, the data are stored on the instrument’s memory and are sent to the connected QIAcuity Software Suite for analysis
Besides system stability and performance reliability improvements, this new version of the control software offers advanced user management and audit trail functionalities. Together with the QIAcuity Software Suite 2.0, the QIAcuity systems now support 21 CFR Part 11 regulations for users working under GMP.
Detailed information is available in the Release Note, which can also be downloaded under section “Operating Software”.
Note: Upgrading the QIAcuity Control Software to version 2.0 will require upgrading the QIAcuity Software Suite to version 2.0.
Important: Please follow the instructions provided in the QIAcuity User Manual for software version 2.0. It is strongly recommended to update the QIAcuity Software Suite first before proceeding with the QIAcuity Control Software!
The QIAcuity Control Software is an integral part of the QIAcuity instrument. It offers a GUI (graphical user interface) for basic functionalities such as plate setup, changing the order of plates to be processed, and monitoring the status of runs in real time. After a run is completed, the data are stored on the instrument’s memory and are sent to the connected QIAcuity Software Suite for analysis.
This new version of the control software includes system stability and performance reliability improvements. We highly recommend to update the control software of your QIAcuity instrument.
Note: The installation requires approximately 90 minutes.
Version 2.0
The QIAcuity Software Suite 2.0 is designed to be installed on a Windows PC that is connected to one or more QIAcuity instruments. The QIAcuity Software Suite enables the user to set up plates, analyze results, and monitor the status of runs in real time. For this configuration, the QIAcuity instrument needs to be connected to a network through Ethernet. Alternatively, a direct cable connection between the QIAcuity and the notebook where the QIAcuity Software Suite is running needs to be established. When connected to a network, up to 10 users may access the QIAcuity Software Suite via a browser installed on the client PC (Windows or Mac).
This new software version now supports 21 CFR Part 11 regulations for users working under GMP. This includes the following:
-Advanced User Management
-Audit Trail
-Signatures
Additional changes compared with the previous software version include the following:
-Usability improvements
-Improved dust detection
-Hyperwell expansion to CNV and GEX analysis
-CSV export functionality of multiple positive partitions for linkage analysis, drop-off assays, etc.
-Raw data export
Important: A direct update of the Software Suite from version 1.1.3 to 2.0.20 is not possible. Please follow the instructions in the User Manual on how to update to version 1.2.18 first before updating the Software Suite to version 2.0.20.
Before you start upgrading to version 1.1.3, please make sure that all plates are imported and visible in the plate overview of the QIAcuity Software Suite.
Caution: Not following the instructions may result in a loss of your previous plate data!
Please contact QIAGEN Technical Services if you are unsure and require technical support.
SHA1 checksum: C60C4EEC349DF1C037796016D32A93EEC19B54A0
Version 2.1
Version 2.1
Beside of improvements and bug fixes, the new features mainly address the GMP requirements by offering additional support for 21 CFR part 11 compliance and get the software ready for future Nanoplate support.
For more information, please refer to the Release Note: QIAcuity Instrument Control Software, Version 2.1.
SHA1 checksum: 0DD41A8DE46D10E81F538ACB71AC184170D39FE0
Version 1.2
The QIAcuity Software Suite 1.2 is designed to be installed on a Windows PC that is connected to one or more QIAcuity instruments. The QIAcuity Software Suite enables the user to set up plates, analyze results, and monitor the status of runs in real time. For this configuration, the QIAcuity instrument needs to be connected to a network through Ethernet. Alternatively, a direct cable connection between the QIAcuity and the notebook where the QIAcuity Software Suite is running needs to be established. When connected to a network, up to 10 users may access the QIAcuity Software Suite via a browser installed on the client PC (Windows or Mac).
The following browsers are supported in the QIAcuity Software Suite:
-Mozilla Firefox (version 64.0.2 or higher)
-Microsoft Edge (version 44.17763.1.0 or higher)
-Google Chrome (version 71.0.3578.98 or higher)
The new QIAcuity Software Suite 1.2 offers a functionality that enables users of the QIAcuity Software 1.1.3 to upgrade to the new version while keeping the library of previously stored plate runs.
Note: If you have exported plates from QIAcuity Software Suite 1.1.3 that you would like to import and use in QIAcuity Software Suite 1.2, you will need to import the plates before upgrading from version 1.1.3 to version 1.2. You may then export the plates again. Future software version starting from QIAcuity Software Suite 2.0 will facilitate import of plates from previous QIAcuity Software Suite versions.
The new improvements are as follows:
-Support for the Nanoplate 8.5k 24-well
-Hyperwell functionality to combine several wells to one combined well for analysis
-Automated plate archiving functionality
-Functionality to show the number of single/double positives in 2D scatterplots
-VPF (Volume Precision Factor) to further improve concentration calculation (see related resources)
-Additional improvements for stabilization and troubleshooting
Version 2.0
The QIAcuity Software Suite 2.0 is designed to be installed on a Windows PC that is connected to one or more QIAcuity instruments. The QIAcuity Software Suite enables the user to set up plates, analyze results, and monitor the status of runs in real time. For this configuration, the QIAcuity instrument needs to be connected to a network through Ethernet. Alternatively, a direct cable connection between the QIAcuity and the notebook where the QIAcuity Software Suite is running needs to be established. When connected to a network, up to 10 users may access the QIAcuity Software Suite via a browser installed on the client PC (Windows or Mac).
This new software version now supports 21 CFR Part 11 regulations for users working under GMP. This includes the following:
-Advanced User Management
-Audit Trail
-Signatures
Additional changes compared with the previous software version include the following:
-Usability improvements
-Improved dust detection
-Hyperwell expansion to CNV and GEX analysis
-CSV export functionality of multiple positive partitions for linkage analysis, drop-off assays, etc.
-Raw data export
Important: A direct update of the Software Suite from version 1.1.3 to 2.0.20 is not possible. Please follow the instructions in the User Manual on how to update to version 1.2.18 first before updating the Software Suite to version 2.0.20.
Before you start upgrading to version 1.1.3, please make sure that all plates are imported and visible in the plate overview of the QIAcuity Software Suite.
Caution: Not following the instructions may result in a loss of your previous plate data!
Please contact QIAGEN Technical Services if you are unsure and require technical support.
SHA1 checksum: C60C4EEC349DF1C037796016D32A93EEC19B54A0
July 2021
The QIAcuity Control Software is an integral part of the QIAcuity instrument. It offers a GUI (graphical user interface) for basic functionalities such as plate setup, changing the order of plates to be processed, and monitoring the status of runs in real time. After a run is completed, the data are stored on the instrument’s memory and are sent to the connected QIAcuity Software Suite for analysis.
This new version of the control software includes system stability and performance reliability improvements. We highly recommend to update the control software of your QIAcuity instrument.
Note: The installation requires approximately 90 minutes.
The QIAcuity Control Software is an integral part of the QIAcuity instrument. It offers a GUI (graphical user interface) for basic functionalities such as plate setup, changing the order of plates to be processed, and monitoring the status of runs in real time. After a run is completed, the data are stored on the instrument’s memory and are sent to the connected QIAcuity Software Suite for analysis
Besides system stability and performance reliability improvements, this new version of the control software offers advanced user management and audit trail functionalities. Together with the QIAcuity Software Suite 2.0, the QIAcuity systems now support 21 CFR Part 11 regulations for users working under GMP.
Detailed information is available in the Release Note, which can also be downloaded under section “Operating Software”.
Note: Upgrading the QIAcuity Control Software to version 2.0 will require upgrading the QIAcuity Software Suite to version 2.0.
Important: Please follow the instructions provided in the QIAcuity User Manual for software version 2.0. It is strongly recommended to update the QIAcuity Software Suite first before proceeding with the QIAcuity Control Software!
Version 2.1
Beside of improvements and bug fixes, the new features mainly address the GMP requirements by offering additional support for 21 CFR part 11 compliance and get the software ready for future Nanoplate support.
For more information, please refer to the Release Note: QIAcuity Instrument Control Software, Version 2.1.
SHA1 checksum: 0DD41A8DE46D10E81F538ACB71AC184170D39FE0
Version 1.2
The QIAcuity Software Suite 1.2 is designed to be installed on a Windows PC that is connected to one or more QIAcuity instruments. The QIAcuity Software Suite enables the user to set up plates, analyze results, and monitor the status of runs in real time. For this configuration, the QIAcuity instrument needs to be connected to a network through Ethernet. Alternatively, a direct cable connection between the QIAcuity and the notebook where the QIAcuity Software Suite is running needs to be established. When connected to a network, up to 10 users may access the QIAcuity Software Suite via a browser installed on the client PC (Windows or Mac).
The following browsers are supported in the QIAcuity Software Suite:
-Mozilla Firefox (version 64.0.2 or higher)
-Microsoft Edge (version 44.17763.1.0 or higher)
-Google Chrome (version 71.0.3578.98 or higher)
The new QIAcuity Software Suite 1.2 offers a functionality that enables users of the QIAcuity Software 1.1.3 to upgrade to the new version while keeping the library of previously stored plate runs.
Note: If you have exported plates from QIAcuity Software Suite 1.1.3 that you would like to import and use in QIAcuity Software Suite 1.2, you will need to import the plates before upgrading from version 1.1.3 to version 1.2. You may then export the plates again. Future software version starting from QIAcuity Software Suite 2.0 will facilitate import of plates from previous QIAcuity Software Suite versions.
The new improvements are as follows:
-Support for the Nanoplate 8.5k 24-well
-Hyperwell functionality to combine several wells to one combined well for analysis
-Automated plate archiving functionality
-Functionality to show the number of single/double positives in 2D scatterplots
-VPF (Volume Precision Factor) to further improve concentration calculation (see related resources)
-Additional improvements for stabilization and troubleshooting
The Volume Precision Factor (VPF) offers a unique feature to secure precision of concentration results obtained from a QIAcuity dPCR run.
In general, Nanoplates provide partitions of fixed sizes that enable a very precise way of sample concentration calculation. Potential variation of partition sizes in Nanoplate batches, caused by different microstructure molding forms, can be addressed by applying the batch specific VPF. Furthermore, the VPF includes well-specific volume information and therefore further increases precision of concentration calculation in each well of the Nanoplates.
After downloading and updating the VPF file within the QIAcuity Software Suite, the VPF is applied automatically to the analysis of a corresponding Nanoplate batch. The VPF file includes information from all available microstructure molding forms and connected Nanoplate batches. It will be stored on the PC where the QIAcuity Software Suite is installed.
Required QIAcuity Software Suite version: Version 1.2 or higher.
Version 2.0
The QIAcuity Software Suite 2.0 is designed to be installed on a Windows PC that is connected to one or more QIAcuity instruments. The QIAcuity Software Suite enables the user to set up plates, analyze results, and monitor the status of runs in real time. For this configuration, the QIAcuity instrument needs to be connected to a network through Ethernet. Alternatively, a direct cable connection between the QIAcuity and the notebook where the QIAcuity Software Suite is running needs to be established. When connected to a network, up to 10 users may access the QIAcuity Software Suite via a browser installed on the client PC (Windows or Mac).
This new software version now supports 21 CFR Part 11 regulations for users working under GMP. This includes the following:
-Advanced User Management
-Audit Trail
-Signatures
Additional changes compared with the previous software version include the following:
-Usability improvements
-Improved dust detection
-Hyperwell expansion to CNV and GEX analysis
-CSV export functionality of multiple positive partitions for linkage analysis, drop-off assays, etc.
-Raw data export
Important: A direct update of the Software Suite from version 1.1.3 to 2.0.20 is not possible. Please follow the instructions in the User Manual on how to update to version 1.2.18 first before updating the Software Suite to version 2.0.20.
Before you start upgrading to version 1.1.3, please make sure that all plates are imported and visible in the plate overview of the QIAcuity Software Suite.
Caution: Not following the instructions may result in a loss of your previous plate data!
Please contact QIAGEN Technical Services if you are unsure and require technical support.
SHA1 checksum: C60C4EEC349DF1C037796016D32A93EEC19B54A0
Version 2.1
Beside of usability improvements and bug fixes, the new features address GMP requirements by offering additional support for 21 CFR part 11 compliance, copy number variation (CNV) analysis, support of future Nanoplates, and improved 1D scatterplot views.
For more information, please refer to the Release Note: QIAcuity Software Suite, Version 2.1
SHA1 checksum: 6D51E273B26FA7D7E0EDA843CB3DCD74B38FA5B1
Version 2.1
Version 2.1
July 2021
FAQ
The plate is designed for a single use run. For example, even if only 30 samples are loaded into the 96-well plate, a whole plate will be sealed by the roller. It can't be unsealed and used for another run. The QIAcuity Software won’t allow to set up a separate experiment for the same nanoplate to avoid that previously processed plates being not partitioned a second time.
The QIAcuity EG PCR Kit should be stored immediately upon receipt at –30 to –15°C in a constant-temperature freezer and protected from light. The QIAcuity EG PCR master mix can also be stored protected from light at 2–8°C. Components are stable for 6 months, unless otherwise indicated on the label.
The QIAcuity Nanoplates does not have expiry date and are stable for at least 1 year when stored at RT.
A standard PCR plate is required to set up dPCR reaction before transferring it to the nanoplate to ensure a proper mixing of the reaction mix before partitioning.
QIAGEN dPCR assays (such as dPCR LNA Mutation Assays) can be found on https://geneglobe.qiagen.com/.
This is not needed. The QIAcuity is equipped with a flexible power supply technology and operates within a range of 100–240V AC, 50/60 Hz, 1500 VA (max).
If you had run a nanoplate for which the installed VPF misses the specific factor, the software will notify you. If you then analyze without the specific VPF, the impact depends on the variation of the partition volume of the new Nanoplate batch compared to the latest. Typically this variation is ±6–7% (approx. 5% CV over the entire plate). The analysis may be repeated after updating the VPF file. After installing the latest VPF and re-analysis of the run, a copy of the plate is generated in the QIAcuity Software Suite including the new results.
QIAGEN master mixes are optimized for nanoplate microfluidics and are recommended to be used with our dPCR system. They also include an optimized reference dye required for proper analysis.
QIAcuity does not support temperature gradient in PCR cycling profile. However, optimization of a dPCR assay can be done by qPCR on a gradient cycler using the dPCR master mix and then transferred from qPCR to dPCR.
QIAGEN offers a complete range of nucleic acid purification systems. These include QIAprep kits for purification of plasmid DNA, QIAamp, and DNeasy kits for purification of genomic DNA, RNeasy kits for purification of total RNA, and the PAXgene Blood RNA System for stabilization and purification of RNA from blood. Phenol and other contaminants can be efficiently removed from crude RNA preps using the RNeasy MinElute Cleanup Kit to clean up and concentrate RNA for sensitive assays. Details about QIAGEN kits for nucleic acid purification can be found at www.qiagen.com.
If you had analyzed your nanoplates without VFP, the impact depends on the variation of the partition volume of the new nanoplate batch compared to the latest. The VPF reduces the CV from approximately 5% to 2%. We recommend to reanalyze results in case the data originated from different wells (e.g., copy number variations or gene expression data sets for which the reference sample was measured in a different well). Results obtained across different plates should also be r-analyzed. A reanalysis is not required for assay data that were analyzed within the same well (e.g., mutation rate determination using two channels within the same well).
In dPCR we measure the absolute concentration of targets at endpoint reaction. Concentrations of unknowns can be determined based on dPCR results observed (number of negatives, number of positives, and partition volume analyzed).
In general, nanoplates provide partitions of fixed sizes that enable a very precise way of sample concentration calculation. If a new molding form is used for nanoplate manufacturing, potential variation of partition sizes can be addressed by applying the molding form-specific VPF. Thus, each time a new molding form is used, a new VPF is created and made available. Currently, the VPF is updated once every 3–6 months.
Nanoplate 8.5K 24-well:12 μl
Nanoplate 8.5K 96-well: 12 μl
The fluorescent signal in the reference channel is measured to determine the number of valid partitions in a well. In addition, differences in the signal intensities between partitions are normalized and the fluorescent signals in the target channels are corrected accordingly.
The VPF provides a set of well-specific and molding form-specific factors used to specify the exact reaction volume of a nanoplate, thus increasing the concentration calculation of each well.
Yes, the report includes a notification if the matching VPF was missing and, therefore, not applied to the analysis. If the matching VPF was applied there is no notification on the report.
The QIAcuity Probe PCR Kit should be stored immediately upon receipt at –30 to –15°C in a constant-temperature freezer and protected from light. The QIAcuity Probe PCR master mix can also be stored protected from light at 2–8°C. Components are stable for 12 months, unless otherwise indicated on the label.
The QIAcuity EG PCR Kit should be stored immediately upon receipt at –30 to –15°C in a constant-temperature freezer and protected from light. The QIAcuity EG PCR master mix can also be stored protected from light at 2–8°C. Components are stable for 6 months, unless otherwise indicated on the label.
The QIAcuity Nanoplates does not have expiry date and are stable for at least 1 year when stored at RT.
Nanoplate 26K 24-well: 40 μl
Nanoplate 8.5K 24-well:12 μl
Nanoplate 8.5K 96-well: 12 μl
All DNA samples used in reaction mixes should show similar quality and quantity, which can easily be assessed using UV spectrophotometry. DNA samples with an average length of ≥20 kb (e.g., genomic DNA purified via spin column with silica membrane) should be fragmented by restriction digestion before partitioning. Enzymatic fragmentation of larger DNA ensures an even distribution of template throughout the QIAcuity Nanoplate, which in turn leads to an accurate and precise quantification.
The instrument software GUI shows error codes including a description and information how to resolve the error. The instrument touchscreen shows an alarm icon in the upper right corner that turns red in case of an instrument failure. Accessing the System Status in the Tool tab allows users to clear errors. Rebooting of the instrument is required to complete the removal of the error. Please do not skip this step. You may always contact QIAGEN Technical Services in case of any question.
The QIAcuity instrument software does not allow to read and process a plate without seal. If you would like to perform a dry run please use sealed plate and set up this plate in the QIAcuity Software Suite.
dPCR master mix can be used in qPCR to optimize sample concentration and/or primer/probe concentration prior to assay transfer from qPCR to dPCR.
The user manual contains instructions on how to perform a regular cleaning and decontamination, and how to replace air filters on the QIAcuity instruments. A regular maintenance reduces the dust in the instrument and therefore minimizes the presence of dust particles on the nanoplate, which might interfere with the plate analysis.
The up-to-date VPF can be downloaded in the resource section of the QIAcuity product webpage. The VPF is compatible with the QIAcuity Software Suite 1.2 and higher. The latest VPF file contains all factors for all existing nanoplate batches. If a nanoplate from a new nanoplate batch is run and the latest version of the VPF was not installed, the software will recognize this and will give a message to install the latest VPF file. Please note that Applying the VPF file cannot be reversed.
The QIAcuity reads emitted fluorescence from the bottom of the plate, which is covered with a foil. For best results, keep the foil clean and avoid damages such as scratches. Also, keep the barcode on the side of the plate clean and intact. Ensure that you wear gloves when working with a plate and do not apply force to it. For a safe handling of the plate, please place the plate into a nanoplate tray.
No. The QIAcuity platform introduces four variations: QIAcuity One 2plex, QIAcuity One 5plex, QIAcuity Four (5plex), and QIAcuity Eight (5plex). All of them have fix channel combination.
Results are stored as part of the plates within the QIAcuity Software Suite. In version 1.1.2, plates can be exported to another file location, for example, an external HDD, and imported again if needed. From version 1.2 onwards, plates can also be archived automatically. To do so, an archive destination has to be defined. Additional information can be found in the user manual.
Both software are designed to be upgraded by users. The user manual includes instructions on how to perform the upgrades; instructions for the QIAcuity Suite Software upgrade can be found at page 38 and instructions for the CSW upgrade on page 67 (QIAcuity User Manual, 03/2021).
