✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
QIAamp DSP Virus Kit
Cat no. / ID. 60704
✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
Features
- Universal purification system compatible with other IVD products
- High-quality viral nucleic acids
- Concentration with 20 µl or 60 µl eluates
- Sample volumes of 500 µl
- Rapid purification and minimal risk of cross-contamination
Product Details
The QIAamp DSP Virus Kit uses well-established and convenient QIAamp technology for simultaneous purification of viral DNA and RNA for in vitro diagnostic use.
Performance
Viral nucleic acids purified using the QIAamp DSP Virus Kit are ready to use in sensitive downstream applications, such as those based on enzymatic amplification, including PCR and RT-PCR.
Principle
The QIAamp DSP Virus Kit uses well-established and convenient QIAamp technology for simultaneous purification of viral DNA and RNA. The QIAamp silica-based membrane binds nucleic acids in the lysed sample, while the rest of the lysate is rapidly removed by vacuum pressure. The bound nucleic acids are efficiently washed to remove contaminants and then eluted in a volume of 20 µl or 60 µl.
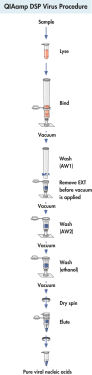
Procedure
See figures
Applications
Viral nucleic acids can be purified from plasma or serum samples. Samples can contain the anticoagulants citrate or EDTA, and can be either fresh, lyophilized, or frozen (provided they were not thawed and refrozen).
The QIAamp DSP Virus Kit provides purification of nucleic acids from a wide range of viruses. The kit is compatible with a wide range of upstream sample collection systems and downstream applications, and therefore can be easily integrated into diagnostic workflows.
Supporting data and figures
QIAamp DSP Virus Kit procedure.
Specifications
| Features | Specifications |
|---|---|
| Applications | Downstream detection procedures in molecular diagnostics, such as PCR |
| Elution volume | 20 µl, 60 µl |
| Main sample type | Serum, plasma |
| CE/FDA/IVD compatible | CE/IVD |
| Format | Sample tubes |
| Processing | Manual (vacuum) |
| Purification of total RNA, miRNA, poly A+ mRNA, DNA or protein | Viral DNA, viral RNA |
| Sample amount | 500 µl |
| Technology | Silica technology |
| Yield | Varies |