✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
therascreen EGFR Pyro Kit (24)
Cat no. / ID. 971480
✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
Features
- Compliance with EU IVD Directive 98/79/EC
- Comprehensive results in real time
- Accurate quantification of mutations in the EGFR gene
- Easy interpretation of complex sequence information
Product Details
Performance
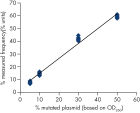
Linearity
Linearity was determined using mixtures of plasmids carrying the wild-type or mutant sequence for the mutations GGC->AGC codon 719, ACG->ATG in codon 790, CTG->CGG in codon 858, and the deletions 2235del15 and 2236del15 in Exon 19 (see figure " Linearity of deletion 2235del15"). The plasmids were mixed in proportions to give four levels of mutation (5, 10, 30, and 50%). Each mixture was analyzed with three different lots of the therascreen EGFR Pyro Kit in three Pyrosequencing runs with three replicates each.
The results were linear within an allowable nonlinearity of 5 % units in the tested range of 5 to 50% mutation level. Similar results were obtained for the mutations GGC->AGC in codon 719, ACG->ATG in codon 790, CTG->CGG in codon 858, and the deletion 2236del15 in Exon 19.
Precision
The precision data allows the determination of the total variability of the assays and was obtained at three different levels by analysis of the above mentioned plasmid mixtures with three replicates each.
Repeatability (intra-assay and inter-batch variability) was calculated based on the data for determination of linearity (three runs on the same day using varying lots of the therascreen EGFR Pyro Kit). Intermediate precision (intra laboratory variability) was determined in three runs within one laboratory on three different days with varying operators, PyroMark Q24 instruments, and lots of the therascreen EGFR Pyro Kit. Reproducibility (inter-laboratory variability) was calculated from two runs each in an internal and external laboratory and using varying lots of the therascreen EGFR Pyro Kit.
Precision estimates are expressed as standard deviation of the measured mutation frequencies in % units. The repeatability, intermediate precision, and reproducibility for the deletion 2235del15 in exon 19 was 0.8–1.2, 0.7–2.9, and 0.7–1.8 % units, respectively, in the measured range of 5 to 50% mutation level. Similar results were obtained for the mutations GGC->AGC in codon 719, ACG->ATG in codon 790, CTG->CGG in codon 858, and the deletion 2236del15 in exon 19.
| % mutated plasmid | Repeatability (Mean, SD) | Intermediate precision (Mean, SD) | Reproducibility (Mean, SD) |
|---|---|---|---|
| 5 | 7.7, 0.8 | 7.4, 0.7 | 7.4, 0.7 |
| 10 | 14.7, 1.1 | 14.5, 1.3 | 14.4, 1.1 |
| 30 | 41.8, 1.2 | 40.0, 2.0 | 41.5, 1.7 |
| 50 | 59.4, 1.0 | 58.2, 2.9 | 60.7, 1.8 |
See figures
Principle
The therascreen EGFR Pyro Kit is used for quantitative measurements of mutations in codons 719, 768, 790, 858, and 861, as well as deletions and complex mutations in exon 19 of the human EGFR gene in real time using Pyrosequencing technology on the PyroMark Q24 System. The EGFR gene encodes the epidermal growth factor receptor (EGFR) protein. Mutations in the tyrosine kinase domain of the EGFR gene can enable tumor growth and progression. EGFR mutations are found in approximately 10% of non-small cell lung cancer incidences in the US and 35% in East Asia. Additionally, EGFR mutations are found in 6% of brain tumors.
The following mutations are detected:
- Exon 18 (719): G719A, G719C, G719S
- Exon 19 (Del): 20 deletions and complex mutations
- Exon 20 (768 and 790): S768I, T790M
- Exon 21 (858–861): L858R, L861Q, L861R
Additional mutations can be detected and analyzed manually.
Procedure
After PCR using primers targeting exons 18, 19, 20, and 21, the amplicons are immobilized on Streptavidin Sepharose High Performance beads. Single-stranded DNA is prepared, and the corresponding sequencing primers anneal to the DNA. The samples are then analyzed on the PyroMark Q24 System using assay setup files and a run setup file.
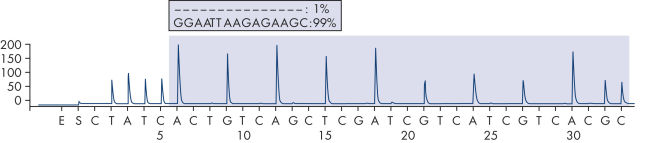
It is recommended to use the EGFR Plug-in Report to analyze the run. This report ensures that the correct LODs are used and different sequences to analyze are automatically used to detect all mutations and deletions. However, the run can also be analyzed using the analysis tool integral to the PyroMark Q24 System (see figures "Pyrogram trace of a normal genotype in codon 719", "Pyrogram trace of a normal genotype in codon 768", " Pyrogram trace of a normal genotype in exon 19", and "Pyrogram trace of a 2235del15 deletion in exon 19"). The "Sequence to Analyze" can then be adjusted for detection different deletions in exon 19 and for rare mutations in the other exons after the run.
See figures
Applications
The therascreen EGFR Pyro Kit enables detection and quantitative measurement of mutations in codons 719, 768, 790, and 858–861, as well as deletions and complex mutations in exon 19 of the human EGFR gene. The kit is intended to be used as an aid to identify cancer patients more likely to benefit from treatment with tyrosine kinase inhibitor therapies.
The following mutations are detected:
- Exon 18 (719): G719A, G719C, G719S<
- Exon 19 (Del): 20 deletions and complex mutations
- Exon 20 (768 and 790): S768I, T790M
- Exon 21 (858–861): L858R, L861Q, L861R
Additional mutations can be detected and analyzed manually.
Supporting data and figures
Pyrogram trace of a normal genotype in exon 19.