QIAGEN Plasmid Kits (Alternative)
For purification of up to 10 mg transfection-grade plasmid or cosmid DNA reviewsubtitle
For purification of up to 10 mg transfection-grade plasmid or cosmid DNA reviewsubtitle

Cat. No. / ID: 12123
QIAGEN Plasmid Kits provide gravity-flow, anion-exchange tips for purification of transfection-grade plasmid DNA. Lysate clearing and isopropanol precipitation are achieved by centrifugation.
The QIAGEN Plasmid Mega Kit (cat. no. 12181) and the QIAGEN Plasmid Giga Kit (cat. no. 12191) can be used with the QIAfilter Mega-Giga Cartridges (cat. no. 19781) as an optional protocol step for rapid clearing of bacterial lysates by filtration instead of centrifugation. See more information regarding principle below.
Test 12123
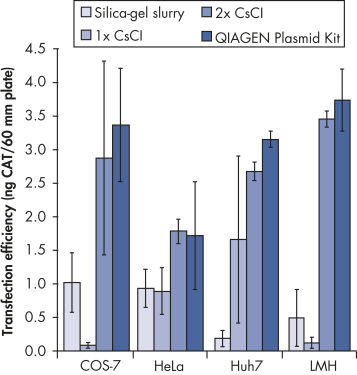
The QIAGEN Plasmid Kits uses gravity-flow QIAGEN anion-exchange tips for efficient purification of plasmid DNA. Up to 10 mg (giga), 2.5 mg (mega), 500 µg (maxi), 100 µg (midi), and 20 µg (mini) high-copy plasmid DNA is purified from culture (culture volumes depend on plasmid copy number, size of insert, host strain, and culture medium). Plasmid DNA purified with QIAGEN Plasmid Kits is highly suitable for use in applications such as transfection (see figure " Transfection efficiency versus plasmid purification method"), cloning, and in vitro transcription.
The unique anion-exchange resin in QIAGEN-tips is developed exclusively for the purification of nucleic acids. Its exceptional separation properties result in DNA purity equivalent or superior to that obtained by two successive rounds of CsCl gradient centrifugation. Prepacked QIAGEN-tips operate by gravity flow and never run dry, minimizing the hands-on time required for plasmid preparation. The entire QIAGEN plasmid purification system avoids the use of toxic substances such as phenol, chloroform, ethidium bromide, and CsCl, minimizing hazard both to the user and the environment.
As an optional protocol step for rapid clearing of bacterial lysates by filtration instead of centrifugation, you can use the QIAfilter Mega-Giga Cartridges (cat. no. 19781), which operate with house vacuum to efficiently clear even large volumes of bacterial lysate with minimal effort. The protocol is available in the handbook.
Features |
Plasmid Mini Kit |
Plasmid Midi Kit |
Plasmid Maxi Kit |
Plasmid Mega Kit |
Plasmid Giga Kit |
|---|---|---|---|---|---|
| Applications | Transfection, cloning, sequencing, capillary sequencing, etc. | Transfection, cloning, sequencing, capillary sequencing, etc. | Transfection, cloning, sequencing, capillary sequencing, etc. | Transfection, cloning, sequencing, capillary sequencing, etc. | Transfection, cloning, sequencing, capillary sequencing, etc. |
| Culture volume/starting material | 3–10 ml culture volume | 25–100 ml culture volume | 100–500 ml culture volume | 500 ml – 2.5 liters culture volume | 2.5–5 liters culture volume |
| Elution volume | Variable | Variable | Variable | Variable | Variable |
| Plasmid type | High-copy, low-copy, cosmid DNA | High-copy, low-copy, cosmid DNA | High-copy, low-copy, cosmid DNA | High-copy, low-copy, cosmid DNA | High-copy, low-copy, cosmid DNA |
| Processing | Manual (gravity flow) | Manual (gravity flow) | Manual (gravity flow) | Manual (gravity flow) | Manual (gravity flow) |
| Sample per run | 1 sample per run | 1 sample per run | 1 sample per run | 1 sample per run | 1 sample per run |
| Technology | Anion-exchange technology | Anion-exchange technology | Anion-exchange technology | Anion-exchange technology | Anion-exchange technology |
| Time per run | 80 min | 150 min | 160 min | 220 min | 320 min |
| Yield | <20 µg | up to 100 µg | <500 µg | <2.5 mg | <10 mg |
With QIAGEN Plasmid Kits, bacterial lysates are cleared by centrifugation. The cleared lysate is then loaded onto the anion-exchange tip where plasmid DNA selectively binds under appropriate low-salt and pH conditions. RNA, proteins, metabolites, and other low-molecular-weight impurities are removed by a medium-salt wash, and pure plasmid DNA is eluted in high-salt buffer (see flowchart " QIAGEN Plasmid Kit procedures"). The DNA is concentrated and desalted by isopropanol precipitation and collected by centrifugation.
Plasmid DNA purified with QIAGEN Plasmid Kits is highly suitable for use in applications, such as:
| Features | Specifications |
|---|---|
| Technology | Anion-exchange technology |
| Culture volume/starting material | 3 ml–5 liters culture volume |
| Yield | <20 µg to <10 mg |
| Processing | Manual (gravity flow) |
| Samples per run (throughput) | 1 sample per run |
| Time per run or prep per run | 80–320 min |
| Applications | Transfection, cloning, sequencing, capillary sequencing etc. |
| Plasmid type | High-copy, low-copy, cosmid DNA |