✓ Traitement automatique des commandes en ligne 24 h/24 7 j/7
✓ Assistance technique et produits pertinente et professionnelle
✓ Commande (ou réapprovisionnement) rapide et fiable
QIAcuity EG PCR Kit (1 ml) icon_0368_ls_gen_eco_friendly-s
N° de réf. / ID. 250111
✓ Traitement automatique des commandes en ligne 24 h/24 7 j/7
✓ Assistance technique et produits pertinente et professionnelle
✓ Commande (ou réapprovisionnement) rapide et fiable
Caractéristiques
- Pour les réactions de PCR digitale à base de colorants comprenant EvaGreen
- Mélange principal à la concentration 3x pour charger plus d’échantillons
- Optimisé pour les applications microfluidiques dans les QIAcuity Nanoplates
- Conforme à REACH
Détails produit
Le QIAcuity EG PCR Kit contient un mélange principal à la concentration 3x, prêt à l’emploi et optimisé pour les applications microfluidiques dans les QIAcuity Nanoplates. Ce kit améliore la spécificité et l’efficacité de la PCR digitale à base de colorants pour assurer une analyse quantitative exacte. Un colorant intercalant EvaGreen se fixe sur l’ADN double brin et augmente l’exactitude quantitative des mesures d’ADNg ou d’ADNc sur les instruments de dPCR QIAcuity.
Ce kit fonctionne en conjonction avec le QIAcuity Digital PCR System et les QIAcuity Nanoplates.
Souhaitez-vous en savoir plus sur ce produit et être contacté par l’un de nos spécialistes de la dPCR ? Inscrivez-vous ici, et nous vous contacterons très prochainement.
Performances
Des performances supérieures
Les mélanges principaux QIAcuity pour la détection avec EvaGreen font appel aux dernières versions d’ADN polymérase QIAGEN de haute qualité. La combinaison unique entre la nouvelle ADN polymérase QuantiNova et la technologie de tampon exclusive et éprouvée de QIAGEN, optimisée pour la microfluidique des nanoplaques, fournit des résultats très homogènes en termes de sensibilité, reproductibilité et efficacité.
Détection à base de colorants avec EvaGreen
Le mélange principal spécial du QIAcuity EG PCR Kit permet d’amplifier et de quantifier avec exactitude l’ADN double brin cible. Il comprend un colorant de référence optimisé qui est nécessaire à l’analyse par dPCR et au dénombrement des partitions analysables dans les nanoplaques. De plus, EvaGreen donne un signal de fluorescence plus élevé que SYBR Green aux mêmes concentrations et assure une efficacité, une spécificité et une sensibilité d’amplification maximales dans la dPCR.
Jusqu’à 100 heures de stabilité des réactions
Les mélanges de PCR QIAcuity peuvent être stockés à 30 °C pendant 100 heures sans compromettre les performances des réactions subséquentes. Cette stabilité exceptionnelle, même après un stockage prolongé à température ambiante en l’absence d’agent refroidissant, rend le QIAcuity EG PCR Kit idéal pour la configuration de réactions et la manipulation de piles de plaques haut débit.
Principe
Le QIAcuity EG PCR Kit permet d’effectuer des analyses d’ADNc ou d’ADNg avec la plus grande spécificité grâce à un nouveau mécanisme hot start médié par anticorps. Aux basses températures, l’ADN polymérase QuantiNova DNA est conservée dans un état inactif par l’anticorps QuantiNova et un nouvel additif, le protecteur QuantiNova qui stabilise le complexe. Cela améliore la rigueur du hot start et prévient l’élongation de dimères d’amorces et d’amorces hybridées de façon non spécifique. Dans les 2 minutes qui suivent l’augmentation de la température à 95 °C, l’anticorps QuantiNova et le protecteur QuantiNova sont dénaturés et l’ADN polymérase QuantiNova est activée, permettant l’amplification par PCR.
Vous trouverez ici une description du principe de la réaction de dPCR sur nanoplaque.
Procédure
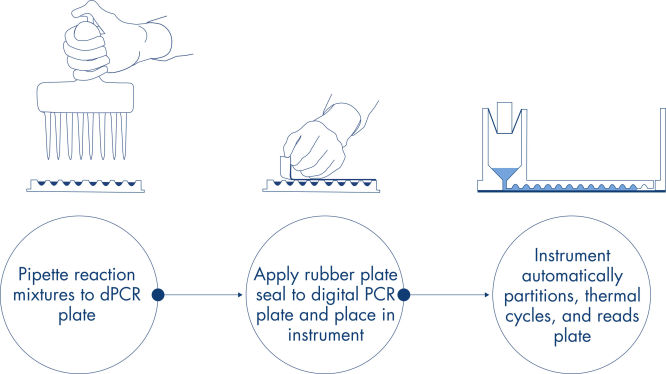
Comme dans les expériences de qPCR, la préparation des échantillons comprend le transfert du mélange principal, des sondes et des amorces sur une nanoplaque de 96 ou 24 puits, suivi de l’addition des échantillons. Le système intègre le partitionnement, le thermocyclage et l’imagerie dans un seul instrument entièrement automatisé qui permet aux utilisateurs de passer de l’échantillon aux résultats en moins de 2 heures. Il est possible d’effectuer l’analyse à l’aide de la suite logicielle, qui donne la concentration de la séquence cible en copies par microlitre et permet le contrôle qualité pour les échantillons positifs ou NTC. Cette analyse peut également être effectuée sur des ordinateurs distants au sein du même réseau local (Local Area Network, LAN).
Applications
Associé avec le QIAcuity Digital PCR System et les QIAcuity Nanoplates, le QIAcuity EG PCR Kit permet d’analyser quantitativement les cibles d’ADNc et l’ADNg pour une utilisation dans les applications telles que :
- La détection des mutations rares
- L’analyse des variations du nombre de copies
- L’analyse de l’expression génique
- La détection des agents pathogènes
- Le génotypage
- La recherche sur les miARN
Données et illustrations utiles
Une méthode sur plaques simple et rapide