- Experiment Configurator
- Discovery & Translational Research
- Diagnostics & Clinical Research
- Human ID & Forensics
- Next-Generation Sequencing
- Instruments & Automation
- Informatics & Data
- Services
- OEM by QIAGEN
- Top Sellers
- New Solutions
Enzymes for molecular biology
Explore high-quality enzymes; now available as individual products.
Products and tools for your targets
Explore targets and pathways in their scientific context, find and customize products to study them, analyze data and plan follow-up studies – all in GeneGlobe.
QuantiFERON-TB Gold Plus
Detect TB infection with confidence.

✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
Cat. No. / ID: 250102
✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
Features
- 适用于单重和最多 5 重反应
- 4x 浓缩预混液,可加载更多样本
- 针对在 QIAcuity Nanoplate 中的微流体使用优化
- 更少的非特异性 PCR 扩增
- 符合 REACH 法规
Product Details
QIAcuity Probe PCR Kit 包含一份用在 QIAcuity Nanoplate 微流体中优化的 4x 浓缩即用型预混液。该试剂盒提高了基于探针法的数字 PCR 的特异性和效率,可提供准确的单重或最多 5 重的定量分析结果。QIAcuity 探针法预混液经过优化,提高特异性,从而可在 QIAcuity dPCR 仪器上准确定量 gDNA 或 cDNA。
该试剂盒应与 QIAcuity Digital PCR System 和 QIAcuity Nanoplate 结合使用。
您想要进一步了解该产品并让我们的 dPCR 专家联系您吗?请在这里登记,我们将很快与您联系。
Performance
卓越的性能
基于水解探针法的 dPCR, QIAcuity 预混液中使用了最新版的 QIAGEN 高品质 DNA 聚合酶。QIAGEN 的专有技术与经过良好验证的专门针对纳米微孔板微流体优化的缓冲液技术及新的 QuantiNova DNA 聚合酶的独特结合,可在灵敏度、重复性及效率等方面获取高度一致的结果。
使用 QIAcuity Probe PCR Kit 可进行基于探针法的单重到五重dPCR检测。
QIAcuity Probe PCR Kit 中的独特预混液可让您在QIAcuity Nanoplate单孔中对最多 5 个丰度大为不同的靶标基因进行准确定量。这可节省时间和金钱,降低所需的样本材料用量。此外,双重或多重 PCR获取的数据与单重 PCR 中获得的数据具有一致性。
反应稳定性可长达 100 个小时——
QIAcuity PCR 混合物可在 30°C 下存放长达 100 个小时,而不会影响后续反应的性能。优异的稳定性——即便在室温不使用冷却剂的情况下,长时间存放后仍保持性能的稳定性,使得 QIAcuity Probe PCR Kit 成为高通量反应体系构建及孔板堆叠处理时的理想选择。
Principle
QIAcuity Probe PCR Kit 采用新型抗体介导的热启动机制,可确保 cDNA 或 gDNA 单重或多重分析时的高特异性。在低温下,QuantiNova DNA 聚合酶在 QuantiNova 抗体和 QuantiNova Guard(一款具有复合物稳定化作用的新型添加剂)的作用下保持在失活状态。这提高了热启动的严格性并防止非特异性退火引物的延伸及引物二聚体的形成。在温度升高到 95°C 的两分钟内,QuantiNova 抗体和 QuantiNova Guard 将发生变性,QuantiNova DNA 聚合酶将被激活,从而启动 PCR 扩增。
有关纳米微孔板中 dPCR 反应的原理说明,请参见这里。
Procedure
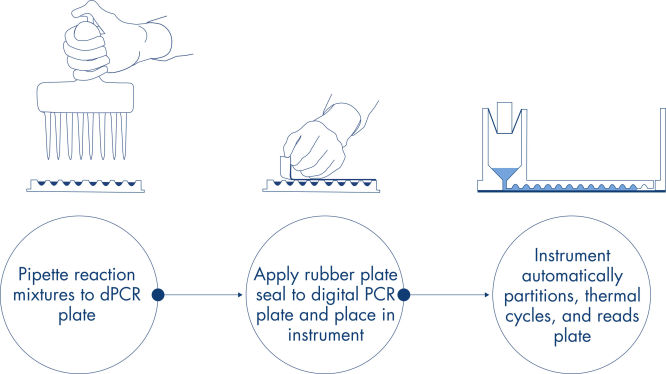
与qPCR 实验一样,反应体系制备包括将预混液、探针和引物转移到一个 96 孔或 24 孔纳米微孔板中,随后添加样本。该系统将分散、循环和成像集成到单个全自动化的仪器上,使用户可在 2 小时内获得结果。您可在 Software Suite 上进行远程分析,该软件可针对您的靶序列及内置的质量控制选项,给出以每微升的拷贝数表示的浓度。该分析还可在同一局域网上的远程计算机上执行。
Applications
QIAcuity Probe PCR Kit 与 QIAcuity Digital PCR System 和 QIAcuity Nanoplate的结合使用可让您在各种应用中进行 cDNA 靶标和 gDNA 定量分析,这些应用包括:
- 罕见突变检测
- 拷贝数变异分析
- 基因表达分析
- 病原体检测
- 基因分型
- miRNA 研究
- 细胞和基因治疗
- 宿主细胞 DNA 残留定量
- 废水监测
Supporting data and figures
简单快速的基于纳米微孔板的工作流程
Resources
The Volume Precision Factor (VPF) offers a unique feature to secure precision of concentration results obtained from a QIAcuity dPCR run.
In general, Nanoplates provide partitions of fixed sizes that enable a very precise way of sample concentration calculation. Potential variation of partition sizes in Nanoplate batches, caused by different microstructure molding forms, can be addressed by applying the batch specific VPF. Furthermore, the VPF includes well-specific volume information and therefore further increases precision of concentration calculation in each well of the Nanoplates.
After downloading and updating the VPF file within the QIAcuity Software Suite, the VPF is applied automatically to the analysis of a corresponding Nanoplate batch. The VPF file includes information from all available microstructure molding forms and connected Nanoplate batches. It will be stored on the PC where the QIAcuity Software Suite is installed.
Required QIAcuity Software Suite version: Version 1.2 or higher.
Version 2.1
Version 2.1
Beside of usability improvements and bug fixes, the new features address GMP requirements by offering additional support for 21 CFR part 11 compliance, copy number variation (CNV) analysis, support of future Nanoplates, and improved 1D scatterplot views.
For more information, please refer to the Release Note: QIAcuity Software Suite, Version 2.1
SHA1 checksum: 6D51E273B26FA7D7E0EDA843CB3DCD74B38FA5B1
The QIAcuity Control Software is an integral part of the QIAcuity instrument. It offers a GUI (graphical user interface) for basic functionalities such as plate setup, changing the order of plates to be processed, and monitoring the status of runs in real time. After a run is completed, the data are stored on the instrument’s memory and are sent to the connected QIAcuity Software Suite for analysis
Besides system stability and performance reliability improvements, this new version of the control software offers advanced user management and audit trail functionalities. Together with the QIAcuity Software Suite 2.0, the QIAcuity systems now support 21 CFR Part 11 regulations for users working under GMP.
Detailed information is available in the Release Note, which can also be downloaded under section “Operating Software”.
Note: Upgrading the QIAcuity Control Software to version 2.0 will require upgrading the QIAcuity Software Suite to version 2.0.
Important: Please follow the instructions provided in the QIAcuity User Manual for software version 2.0. It is strongly recommended to update the QIAcuity Software Suite first before proceeding with the QIAcuity Control Software!
The QIAcuity Control Software is an integral part of the QIAcuity instrument. It offers a GUI (graphical user interface) for basic functionalities such as plate setup, changing the order of plates to be processed, and monitoring the status of runs in real time. After a run is completed, the data are stored on the instrument’s memory and are sent to the connected QIAcuity Software Suite for analysis.
This new version of the control software includes system stability and performance reliability improvements. We highly recommend to update the control software of your QIAcuity instrument.
Note: The installation requires approximately 90 minutes.
Version 2.0
The QIAcuity Software Suite 2.0 is designed to be installed on a Windows PC that is connected to one or more QIAcuity instruments. The QIAcuity Software Suite enables the user to set up plates, analyze results, and monitor the status of runs in real time. For this configuration, the QIAcuity instrument needs to be connected to a network through Ethernet. Alternatively, a direct cable connection between the QIAcuity and the notebook where the QIAcuity Software Suite is running needs to be established. When connected to a network, up to 10 users may access the QIAcuity Software Suite via a browser installed on the client PC (Windows or Mac).
This new software version now supports 21 CFR Part 11 regulations for users working under GMP. This includes the following:
-Advanced User Management
-Audit Trail
-Signatures
Additional changes compared with the previous software version include the following:
-Usability improvements
-Improved dust detection
-Hyperwell expansion to CNV and GEX analysis
-CSV export functionality of multiple positive partitions for linkage analysis, drop-off assays, etc.
-Raw data export
Important: A direct update of the Software Suite from version 1.1.3 to 2.0.20 is not possible. Please follow the instructions in the User Manual on how to update to version 1.2.18 first before updating the Software Suite to version 2.0.20.
Before you start upgrading to version 1.1.3, please make sure that all plates are imported and visible in the plate overview of the QIAcuity Software Suite.
Caution: Not following the instructions may result in a loss of your previous plate data!
Please contact QIAGEN Technical Services if you are unsure and require technical support.
SHA1 checksum: C60C4EEC349DF1C037796016D32A93EEC19B54A0
Version 2.1
Version 2.1
Beside of improvements and bug fixes, the new features mainly address the GMP requirements by offering additional support for 21 CFR part 11 compliance and get the software ready for future Nanoplate support.
For more information, please refer to the Release Note: QIAcuity Instrument Control Software, Version 2.1.
SHA1 checksum: 0DD41A8DE46D10E81F538ACB71AC184170D39FE0
Version 1.2
The QIAcuity Software Suite 1.2 is designed to be installed on a Windows PC that is connected to one or more QIAcuity instruments. The QIAcuity Software Suite enables the user to set up plates, analyze results, and monitor the status of runs in real time. For this configuration, the QIAcuity instrument needs to be connected to a network through Ethernet. Alternatively, a direct cable connection between the QIAcuity and the notebook where the QIAcuity Software Suite is running needs to be established. When connected to a network, up to 10 users may access the QIAcuity Software Suite via a browser installed on the client PC (Windows or Mac).
The following browsers are supported in the QIAcuity Software Suite:
-Mozilla Firefox (version 64.0.2 or higher)
-Microsoft Edge (version 44.17763.1.0 or higher)
-Google Chrome (version 71.0.3578.98 or higher)
The new QIAcuity Software Suite 1.2 offers a functionality that enables users of the QIAcuity Software 1.1.3 to upgrade to the new version while keeping the library of previously stored plate runs.
Note: If you have exported plates from QIAcuity Software Suite 1.1.3 that you would like to import and use in QIAcuity Software Suite 1.2, you will need to import the plates before upgrading from version 1.1.3 to version 1.2. You may then export the plates again. Future software version starting from QIAcuity Software Suite 2.0 will facilitate import of plates from previous QIAcuity Software Suite versions.
The new improvements are as follows:
-Support for the Nanoplate 8.5k 24-well
-Hyperwell functionality to combine several wells to one combined well for analysis
-Automated plate archiving functionality
-Functionality to show the number of single/double positives in 2D scatterplots
-VPF (Volume Precision Factor) to further improve concentration calculation (see related resources)
-Additional improvements for stabilization and troubleshooting
Version 2.0
The QIAcuity Software Suite 2.0 is designed to be installed on a Windows PC that is connected to one or more QIAcuity instruments. The QIAcuity Software Suite enables the user to set up plates, analyze results, and monitor the status of runs in real time. For this configuration, the QIAcuity instrument needs to be connected to a network through Ethernet. Alternatively, a direct cable connection between the QIAcuity and the notebook where the QIAcuity Software Suite is running needs to be established. When connected to a network, up to 10 users may access the QIAcuity Software Suite via a browser installed on the client PC (Windows or Mac).
This new software version now supports 21 CFR Part 11 regulations for users working under GMP. This includes the following:
-Advanced User Management
-Audit Trail
-Signatures
Additional changes compared with the previous software version include the following:
-Usability improvements
-Improved dust detection
-Hyperwell expansion to CNV and GEX analysis
-CSV export functionality of multiple positive partitions for linkage analysis, drop-off assays, etc.
-Raw data export
Important: A direct update of the Software Suite from version 1.1.3 to 2.0.20 is not possible. Please follow the instructions in the User Manual on how to update to version 1.2.18 first before updating the Software Suite to version 2.0.20.
Before you start upgrading to version 1.1.3, please make sure that all plates are imported and visible in the plate overview of the QIAcuity Software Suite.
Caution: Not following the instructions may result in a loss of your previous plate data!
Please contact QIAGEN Technical Services if you are unsure and require technical support.
SHA1 checksum: C60C4EEC349DF1C037796016D32A93EEC19B54A0
July 2021
The QIAcuity Control Software is an integral part of the QIAcuity instrument. It offers a GUI (graphical user interface) for basic functionalities such as plate setup, changing the order of plates to be processed, and monitoring the status of runs in real time. After a run is completed, the data are stored on the instrument’s memory and are sent to the connected QIAcuity Software Suite for analysis.
This new version of the control software includes system stability and performance reliability improvements. We highly recommend to update the control software of your QIAcuity instrument.
Note: The installation requires approximately 90 minutes.
The QIAcuity Control Software is an integral part of the QIAcuity instrument. It offers a GUI (graphical user interface) for basic functionalities such as plate setup, changing the order of plates to be processed, and monitoring the status of runs in real time. After a run is completed, the data are stored on the instrument’s memory and are sent to the connected QIAcuity Software Suite for analysis
Besides system stability and performance reliability improvements, this new version of the control software offers advanced user management and audit trail functionalities. Together with the QIAcuity Software Suite 2.0, the QIAcuity systems now support 21 CFR Part 11 regulations for users working under GMP.
Detailed information is available in the Release Note, which can also be downloaded under section “Operating Software”.
Note: Upgrading the QIAcuity Control Software to version 2.0 will require upgrading the QIAcuity Software Suite to version 2.0.
Important: Please follow the instructions provided in the QIAcuity User Manual for software version 2.0. It is strongly recommended to update the QIAcuity Software Suite first before proceeding with the QIAcuity Control Software!
Version 2.1
Beside of improvements and bug fixes, the new features mainly address the GMP requirements by offering additional support for 21 CFR part 11 compliance and get the software ready for future Nanoplate support.
For more information, please refer to the Release Note: QIAcuity Instrument Control Software, Version 2.1.
SHA1 checksum: 0DD41A8DE46D10E81F538ACB71AC184170D39FE0
Version 1.2
The QIAcuity Software Suite 1.2 is designed to be installed on a Windows PC that is connected to one or more QIAcuity instruments. The QIAcuity Software Suite enables the user to set up plates, analyze results, and monitor the status of runs in real time. For this configuration, the QIAcuity instrument needs to be connected to a network through Ethernet. Alternatively, a direct cable connection between the QIAcuity and the notebook where the QIAcuity Software Suite is running needs to be established. When connected to a network, up to 10 users may access the QIAcuity Software Suite via a browser installed on the client PC (Windows or Mac).
The following browsers are supported in the QIAcuity Software Suite:
-Mozilla Firefox (version 64.0.2 or higher)
-Microsoft Edge (version 44.17763.1.0 or higher)
-Google Chrome (version 71.0.3578.98 or higher)
The new QIAcuity Software Suite 1.2 offers a functionality that enables users of the QIAcuity Software 1.1.3 to upgrade to the new version while keeping the library of previously stored plate runs.
Note: If you have exported plates from QIAcuity Software Suite 1.1.3 that you would like to import and use in QIAcuity Software Suite 1.2, you will need to import the plates before upgrading from version 1.1.3 to version 1.2. You may then export the plates again. Future software version starting from QIAcuity Software Suite 2.0 will facilitate import of plates from previous QIAcuity Software Suite versions.
The new improvements are as follows:
-Support for the Nanoplate 8.5k 24-well
-Hyperwell functionality to combine several wells to one combined well for analysis
-Automated plate archiving functionality
-Functionality to show the number of single/double positives in 2D scatterplots
-VPF (Volume Precision Factor) to further improve concentration calculation (see related resources)
-Additional improvements for stabilization and troubleshooting
The Volume Precision Factor (VPF) offers a unique feature to secure precision of concentration results obtained from a QIAcuity dPCR run.
In general, Nanoplates provide partitions of fixed sizes that enable a very precise way of sample concentration calculation. Potential variation of partition sizes in Nanoplate batches, caused by different microstructure molding forms, can be addressed by applying the batch specific VPF. Furthermore, the VPF includes well-specific volume information and therefore further increases precision of concentration calculation in each well of the Nanoplates.
After downloading and updating the VPF file within the QIAcuity Software Suite, the VPF is applied automatically to the analysis of a corresponding Nanoplate batch. The VPF file includes information from all available microstructure molding forms and connected Nanoplate batches. It will be stored on the PC where the QIAcuity Software Suite is installed.
Required QIAcuity Software Suite version: Version 1.2 or higher.
Version 2.0
The QIAcuity Software Suite 2.0 is designed to be installed on a Windows PC that is connected to one or more QIAcuity instruments. The QIAcuity Software Suite enables the user to set up plates, analyze results, and monitor the status of runs in real time. For this configuration, the QIAcuity instrument needs to be connected to a network through Ethernet. Alternatively, a direct cable connection between the QIAcuity and the notebook where the QIAcuity Software Suite is running needs to be established. When connected to a network, up to 10 users may access the QIAcuity Software Suite via a browser installed on the client PC (Windows or Mac).
This new software version now supports 21 CFR Part 11 regulations for users working under GMP. This includes the following:
-Advanced User Management
-Audit Trail
-Signatures
Additional changes compared with the previous software version include the following:
-Usability improvements
-Improved dust detection
-Hyperwell expansion to CNV and GEX analysis
-CSV export functionality of multiple positive partitions for linkage analysis, drop-off assays, etc.
-Raw data export
Important: A direct update of the Software Suite from version 1.1.3 to 2.0.20 is not possible. Please follow the instructions in the User Manual on how to update to version 1.2.18 first before updating the Software Suite to version 2.0.20.
Before you start upgrading to version 1.1.3, please make sure that all plates are imported and visible in the plate overview of the QIAcuity Software Suite.
Caution: Not following the instructions may result in a loss of your previous plate data!
Please contact QIAGEN Technical Services if you are unsure and require technical support.
SHA1 checksum: C60C4EEC349DF1C037796016D32A93EEC19B54A0
Version 2.1
Beside of usability improvements and bug fixes, the new features address GMP requirements by offering additional support for 21 CFR part 11 compliance, copy number variation (CNV) analysis, support of future Nanoplates, and improved 1D scatterplot views.
For more information, please refer to the Release Note: QIAcuity Software Suite, Version 2.1
SHA1 checksum: 6D51E273B26FA7D7E0EDA843CB3DCD74B38FA5B1
Version 2.1
Version 2.1
July 2021
FAQ
The plate is designed for a single use run. For example, even if only 30 samples are loaded into the 96-well plate, a whole plate will be sealed by the roller. It can't be unsealed and used for another run. The QIAcuity Software won’t allow to set up a separate experiment for the same nanoplate to avoid that previously processed plates being not partitioned a second time.
The QIAcuity EG PCR Kit should be stored immediately upon receipt at –30 to –15°C in a constant-temperature freezer and protected from light. The QIAcuity EG PCR master mix can also be stored protected from light at 2–8°C. Components are stable for 6 months, unless otherwise indicated on the label.
The QIAcuity Nanoplates does not have expiry date and are stable for at least 1 year when stored at RT.
A standard PCR plate is required to set up dPCR reaction before transferring it to the nanoplate to ensure a proper mixing of the reaction mix before partitioning.
QIAGEN dPCR assays (such as dPCR LNA Mutation Assays) can be found on https://geneglobe.qiagen.com/.
This is not needed. The QIAcuity is equipped with a flexible power supply technology and operates within a range of 100–240V AC, 50/60 Hz, 1500 VA (max).
If you had run a nanoplate for which the installed VPF misses the specific factor, the software will notify you. If you then analyze without the specific VPF, the impact depends on the variation of the partition volume of the new Nanoplate batch compared to the latest. Typically this variation is ±6–7% (approx. 5% CV over the entire plate). The analysis may be repeated after updating the VPF file. After installing the latest VPF and re-analysis of the run, a copy of the plate is generated in the QIAcuity Software Suite including the new results.
QIAGEN master mixes are optimized for nanoplate microfluidics and are recommended to be used with our dPCR system. They also include an optimized reference dye required for proper analysis.
QIAcuity does not support temperature gradient in PCR cycling profile. However, optimization of a dPCR assay can be done by qPCR on a gradient cycler using the dPCR master mix and then transferred from qPCR to dPCR.
QIAGEN offers a complete range of nucleic acid purification systems. These include QIAprep kits for purification of plasmid DNA, QIAamp, and DNeasy kits for purification of genomic DNA, RNeasy kits for purification of total RNA, and the PAXgene Blood RNA System for stabilization and purification of RNA from blood. Phenol and other contaminants can be efficiently removed from crude RNA preps using the RNeasy MinElute Cleanup Kit to clean up and concentrate RNA for sensitive assays. Details about QIAGEN kits for nucleic acid purification can be found at www.qiagen.com.
If you had analyzed your nanoplates without VFP, the impact depends on the variation of the partition volume of the new nanoplate batch compared to the latest. The VPF reduces the CV from approximately 5% to 2%. We recommend to reanalyze results in case the data originated from different wells (e.g., copy number variations or gene expression data sets for which the reference sample was measured in a different well). Results obtained across different plates should also be r-analyzed. A reanalysis is not required for assay data that were analyzed within the same well (e.g., mutation rate determination using two channels within the same well).
In dPCR we measure the absolute concentration of targets at endpoint reaction. Concentrations of unknowns can be determined based on dPCR results observed (number of negatives, number of positives, and partition volume analyzed).
In general, nanoplates provide partitions of fixed sizes that enable a very precise way of sample concentration calculation. If a new molding form is used for nanoplate manufacturing, potential variation of partition sizes can be addressed by applying the molding form-specific VPF. Thus, each time a new molding form is used, a new VPF is created and made available. Currently, the VPF is updated once every 3–6 months.
Nanoplate 8.5K 24-well:12 μl
Nanoplate 8.5K 96-well: 12 μl
The fluorescent signal in the reference channel is measured to determine the number of valid partitions in a well. In addition, differences in the signal intensities between partitions are normalized and the fluorescent signals in the target channels are corrected accordingly.
The VPF provides a set of well-specific and molding form-specific factors used to specify the exact reaction volume of a nanoplate, thus increasing the concentration calculation of each well.
Yes, the report includes a notification if the matching VPF was missing and, therefore, not applied to the analysis. If the matching VPF was applied there is no notification on the report.
The QIAcuity Probe PCR Kit should be stored immediately upon receipt at –30 to –15°C in a constant-temperature freezer and protected from light. The QIAcuity Probe PCR master mix can also be stored protected from light at 2–8°C. Components are stable for 12 months, unless otherwise indicated on the label.
The QIAcuity EG PCR Kit should be stored immediately upon receipt at –30 to –15°C in a constant-temperature freezer and protected from light. The QIAcuity EG PCR master mix can also be stored protected from light at 2–8°C. Components are stable for 6 months, unless otherwise indicated on the label.
The QIAcuity Nanoplates does not have expiry date and are stable for at least 1 year when stored at RT.
Nanoplate 26K 24-well: 40 μl
Nanoplate 8.5K 24-well:12 μl
Nanoplate 8.5K 96-well: 12 μl
All DNA samples used in reaction mixes should show similar quality and quantity, which can easily be assessed using UV spectrophotometry. DNA samples with an average length of ≥20 kb (e.g., genomic DNA purified via spin column with silica membrane) should be fragmented by restriction digestion before partitioning. Enzymatic fragmentation of larger DNA ensures an even distribution of template throughout the QIAcuity Nanoplate, which in turn leads to an accurate and precise quantification.
The instrument software GUI shows error codes including a description and information how to resolve the error. The instrument touchscreen shows an alarm icon in the upper right corner that turns red in case of an instrument failure. Accessing the System Status in the Tool tab allows users to clear errors. Rebooting of the instrument is required to complete the removal of the error. Please do not skip this step. You may always contact QIAGEN Technical Services in case of any question.
The QIAcuity instrument software does not allow to read and process a plate without seal. If you would like to perform a dry run please use sealed plate and set up this plate in the QIAcuity Software Suite.
dPCR master mix can be used in qPCR to optimize sample concentration and/or primer/probe concentration prior to assay transfer from qPCR to dPCR.
The user manual contains instructions on how to perform a regular cleaning and decontamination, and how to replace air filters on the QIAcuity instruments. A regular maintenance reduces the dust in the instrument and therefore minimizes the presence of dust particles on the nanoplate, which might interfere with the plate analysis.
The up-to-date VPF can be downloaded in the resource section of the QIAcuity product webpage. The VPF is compatible with the QIAcuity Software Suite 1.2 and higher. The latest VPF file contains all factors for all existing nanoplate batches. If a nanoplate from a new nanoplate batch is run and the latest version of the VPF was not installed, the software will recognize this and will give a message to install the latest VPF file. Please note that Applying the VPF file cannot be reversed.
The QIAcuity reads emitted fluorescence from the bottom of the plate, which is covered with a foil. For best results, keep the foil clean and avoid damages such as scratches. Also, keep the barcode on the side of the plate clean and intact. Ensure that you wear gloves when working with a plate and do not apply force to it. For a safe handling of the plate, please place the plate into a nanoplate tray.
No. The QIAcuity platform introduces four variations: QIAcuity One 2plex, QIAcuity One 5plex, QIAcuity Four (5plex), and QIAcuity Eight (5plex). All of them have fix channel combination.
Results are stored as part of the plates within the QIAcuity Software Suite. In version 1.1.2, plates can be exported to another file location, for example, an external HDD, and imported again if needed. From version 1.2 onwards, plates can also be archived automatically. To do so, an archive destination has to be defined. Additional information can be found in the user manual.
Both software are designed to be upgraded by users. The user manual includes instructions on how to perform the upgrades; instructions for the QIAcuity Suite Software upgrade can be found at page 38 and instructions for the CSW upgrade on page 67 (QIAcuity User Manual, 03/2021).
