HotStarTaq Master Mix Kit
高特异性扩增,用于多种PCR分析
高特异性扩增,用于多种PCR分析
Cat. No. / ID: 203443
每一批HotStarTaq Master Mix Kit都经过全方位的质量控制测试,包括:严格的PCR特异性和可重复性分析,即使低拷贝的靶分子也可扩增。通过检测,HotStarTaq DNA Polymerase优于其它供应商提供的试剂盒,确保高度特异性和在热启动PCR中的卓越表现(参见" Higher specificity with different primer–template systems"、" Superior performance"和表格)。该试剂盒提供的新型PCR缓冲液使得在多种PCR条件下都维持高特异性,无需优化(参见" Tolerance to variable magnesium concentration")。
高度特异性配合简单的操作使HotStarTaq Master Mix Kit适用于复杂的基因组或cDNA模板(参见" Effect of hot start on RT-PCR performance")、多重引物对(参见" Specific amplification in multiplex PCR")、从珍贵样本或低拷贝靶分子中抽提的模板(参见" Highly sensitive single-cell PCR")。该试剂盒还适用于大量样本的扩增,诸如基因筛查等项目。
| HotStarTaq DNA Polymerase | Supplier AII提供的热启动酶 | 抗体介导 | 手动 | 蜡屏障 | |
|---|---|---|---|---|---|
| 特异性扩增 | ++ | + | + | +/– | +/– |
| PCR优化需求 | ++ | +/– | +/– | – | – |
| 易用性 | ++ | ++ | + | – | – |
浓度:5 units/µl
重组酶:是
底物类似物:dNTP、ddNTP、dUTP、biotin-11-dUTP、DIG-11-dUTP、fluorescent-dNTP/ddNTP
延伸速度:72°C条件下2–4 kb/min
酶半衰期:97°C条件下10 min,94°C条件下60 min
扩增效率:≥105倍
5'–>3'外切酶活性:是
额外添加A:是
3'–>5' 外切酶活性:否
核酸酶污染:否
蛋白酶污染:否
RNases污染:否
自启活性:否
HotStarTaq Master Mix是一种即用型预混液,包含HotStarTaq DNA Polymerase、QIAGEN PCR Buffer和dNTPs。HotStarTaq DNA Polymerase是一种经修饰的Taq DNA Polymerase,确保热启动PCR的高特异性。
HotStarTaq DNA Polymerase以非活性形式提供,常温下没有聚合酶活性。避免PCR构建和循环初始阶段低温条件下非特异性引物的延伸和引物二聚体的形成(参见" Superior performance in hot-start PCR"和" Higher specificity with different primer–template systems")。95°C条件下孵育15分钟即可激活HotStarTaq DNA Polymerase,这个步骤可整合入已有的热循环程序中。
QIAGEN PCR Buffer通过提高每个PCR扩增退火过程特异性引物的结合比例,确保PCR每个循环特异性扩增(参见" Increased specificity of primer annealing")。独特的KCl和(NH4)2SO4平衡组合,使得该缓冲液与传统的PCR缓冲液相比在很宽的温度和Mg2+浓度范围内都有严格的引物退火条件和高度的特异性,无需进行耗时的优化(参见" Tolerance to variable magnesium concentration")。
HotStarTaq Master Mix Kit适合多种应用,包括各种富有挑战性的研究,如各种扩增应用:
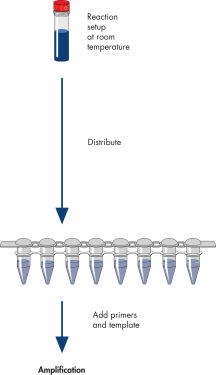
The HotStarTaq procedure is fast and easy for maximum convenience.
| Features | Specifications |
|---|---|
| ApplicationsZH | PCR, RT-PCR, Complex genomic templates, very low-copy targets |
| Real-time or endpoint | Endpoint |
| Mastermix | Yes |
| Enzyme activity | 5'-> 3' exonuclease activity |
| Sample/target type | Genomic DNA and cDNA |
| Single or multiplex | Single |
| Reaction type | PCR amplification |
| With/without hotstart | With hotstart |